
Global Molecular Diagnostics Market Size, Trends & Analysis - Forecasts to 2027 By Product (Instruments, Consumables), By Technology (PCR [Standard PCR, rtPCR], In Situ Hybridization, Chips & Microarrays, Mass Spectrometry (MS), Sequencing), By Application (Infectious Disease [COVID-19, Methicillin-resistant Staphylococcus Aureus (MRSA), Clostridium Difficile, Vancomycin-resistant Enterococci (VRE), Carbapenem-resistant Bacteria, Flu, Respiratory Syncytial Virus (RSV), Candida, Tuberculosis & Drug-resistant TB, Meningitis, Gastrointestinal Panel Testing, Chlamydia, Gonorrhea, HIV, Hepatitis C, Hepatitis B], Oncology [Breast Cancer, Prostate Cancer, Colorectal Cancer, Cervical Cancer, Kidney Cancer, Liver Cancer, Blood Cancer, Lung Cancer], Neurological Disease, Cardiovascular Disease), By End-User (Hospitals & Clinics, Laboratories, Diagnostic Centers, Home Testing), By Region (North America, Asia Pacific, Central & South America, Europe, and the Middle East & Africa), End-User Landscape Analysis, Company Market Share Analysis, and Competitor Analysis
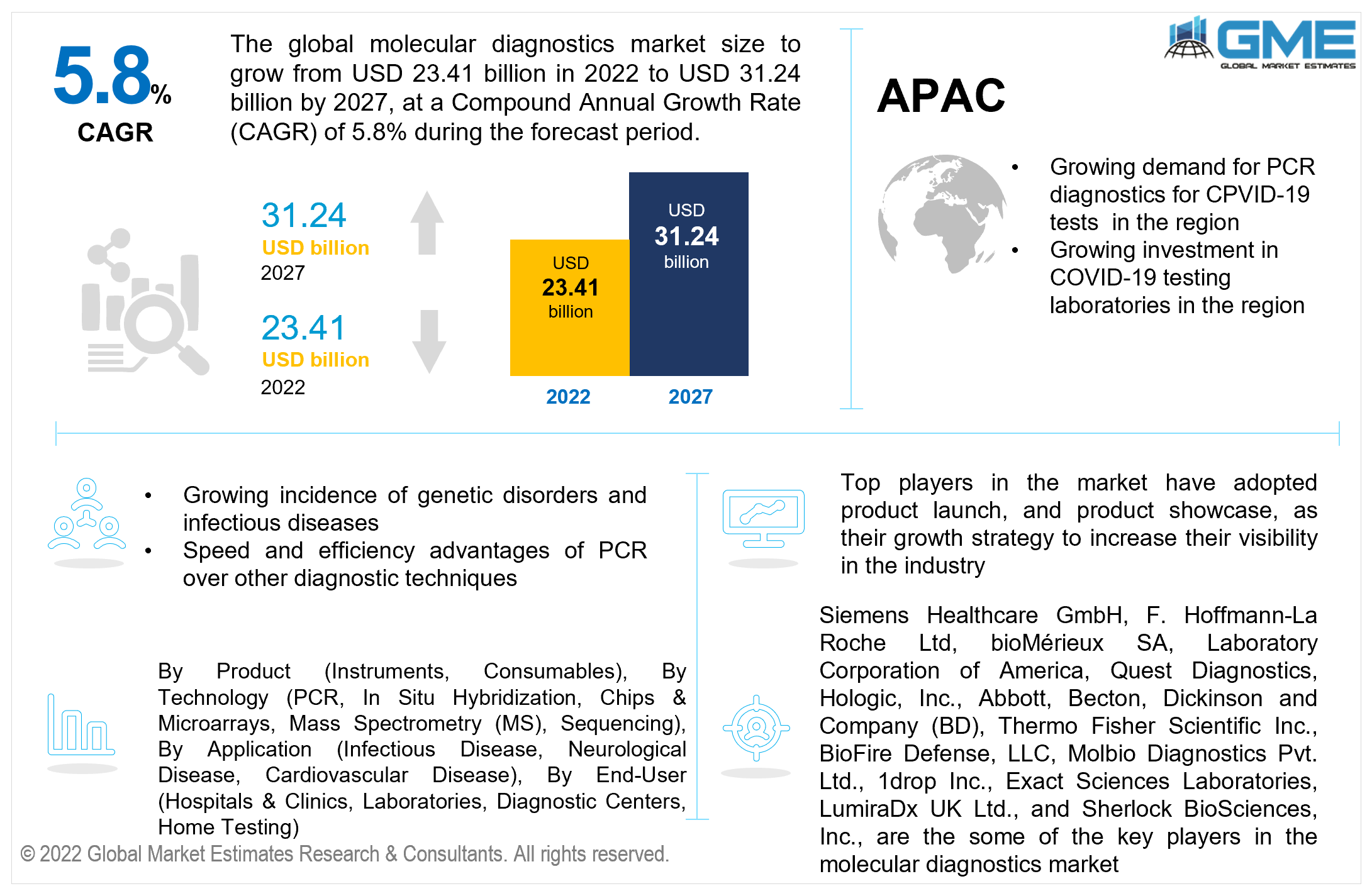
The Global Molecular Diagnostics Market is projected to grow from USD 23.41 billion in 2022 to USD 31.24 billion at a CAGR value of 5.8% from 2022 to 2027.
The identification of individual biologic molecules is the most basic function of molecular diagnostics. The development of user-friendly molecular diagnostic tools is largely responsible for the rise of practical applications of molecular diagnostics in fields such as oncology, cardiology, infectious disease treatments, and neurological disorders, among others.
The advent of PCR (polymerase chain reaction) resulted in a huge breakthrough in clinical DNA testing. PCR-based diagnostic procedures require minimal and less complex instruments and do not place a heavy burden on the infrastructure. These diagnostic techniques require minuscule amounts of biological material and can be easily incorporated within existing laboratory procedures, thereby promoting their rapid adoption. Various conventional tests have been replaced by molecular diagnostic techniques in many areas of medicine, including oncology, infectious diseases, medicinal chemistry, and therapeutic genomics. Molecular diagnostic testing will continue to increase the precision and speed with which diagnostic technicians can detect microbial pathogens or study a patient's genes, and it is becoming an essential piece of patient-tailored treatment and medicines.
Diagnostics are an important element of the healthcare sector, with test findings influencing every step of the patient's journey. Year-on-year improvements in requests for diagnostic tests suggest rising demand for healthcare services, yet these tests are frequently requested late in the disease progression. Early diagnosis, according to clinical evidence, leads to improved health outcomes and is cost-effective. Molecular diagnostics has showcased its potential to help promote timely diagnosis to improve the quality of life of patients. The growing demand for early diagnostics and the need for cost-effective diagnostic techniques are expected to drive the market during the forecast period.
Many molecular diagnostic tests' capacity to successfully demonstrate the presence of the target material in low concentration samples has aided molecular diagnostics' growing popularity. Biomarkers are becoming a more prominent focus of health research, alongside research on molecular markers of distinct biological conditions. New molecular diagnostic procedures are being driven by advances in molecular biology, genomics, and proteomics. Biomarkers can lead to an earlier or conclusive diagnosis, diagnosing at-risk patients, delivering more accurate prognoses, fine-tuning treatment selection, and administering individualized medicine.
The growing demand for molecular diagnostics in orthopedic infection detection is expected to further increase the demand for molecular diagnostics. Despite the presence of orthopedic infections that might not be culturable or have poor virulence, PCR-based techniques have been proven to be useful in discovering bacterial DNA or RNA, which can help determine pathogenic species and medication resistance for applications in orthopedic treatment strategies. These procedures are largely being used to determine the amount of live bacteria present to accurately determine the infection. The identification of specific host proteins in synovial fluid, including cytokines and antimicrobial peptides, is another promising new molecular diagnostic procedure for detecting orthopedic infections.
The market is expected to be hampered by the availability of historically employed alternative diagnostic procedures. The need for upskilling laboratory staff to utilize molecular diagnostic instruments, the need for skilled professionals, the high cost of devices, and the limitations such as false positives and sample contamination are also expected to challenge the growth of the market.
The need for RT-PCR tests has skyrocketed as a result of the COVID-19 pandemic. As RT-PCR testing allows for rapid testing of the population with excellent precision, they have become the gold standard for conducting COVID-19 tests. The molecular diagnostics market has seen rapid growth in the last few years owing to mandatory RT-PCR test results for traveling. As COVID regulations are eased and vaccination rates rise, the strong demand for PCR tests is projected to decline. The market will increase considerably over the forecast period due to the application of PCR testing in numerous other applications and its widespread utilization to combat COVID.
The crisis between Russia and Ukraine will have a considerable impact on the demand for PCR tests within both these nations. The ability of Ukrainian diagnostic laboratories to conduct PCR tests for COVID-19 tests has been severely impeded by the presence of Russian hostiles in Ukraine. Only 2.5 percent to 3% of laboratories capable of doing PCR testing are doing so, according to estimates. Companies all around the world are voluntarily restricting trade with Russia, which will make it difficult to import the consumables needed to conduct molecular diagnostic testing in the country.
The instruments segment is expected to be the largest molecular diagnostics market segment based on the product. The high cost of molecular diagnostic instruments, frequent updates to existing infrastructure, and the growing adoption of molecular diagnostics in laboratories have led to the large demand for instruments.
The instruments segment is also expected to be the fastest-growing segment in the market. The growing demand for RT-PCR tests for COVID tests is increasing the adoption of molecular diagnostics instruments, and increased outsourcing of laboratory functions by hospitals and clinics is expected to increase the demand for molecular diagnostic instruments.
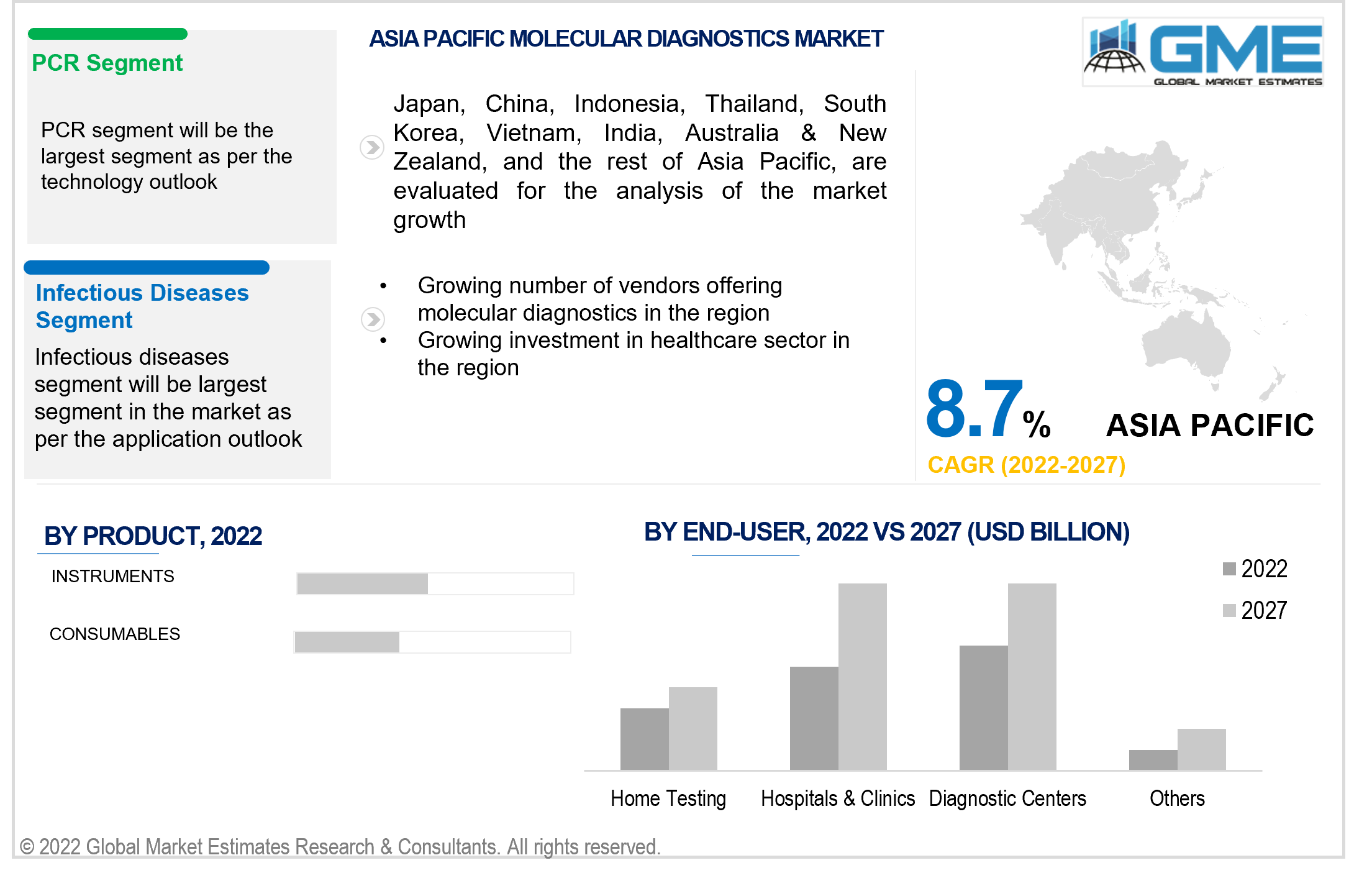
The PCR segment is expected to be the largest molecular diagnostics market segment based on the technology. A large number of applications for PCR in various fields of medicine such as oncology, genomics, cardiology, and neurology, among others have led to the growing adoption of PCR diagnostic tests. The rapid adoption of PCR tests during the COVID pandemic has also played a crucial role in the heavy demand for PCR testing.
The chips and microarrays segment is also expected to be the fastest-growing segment in the market. Technological advancements in chips and microarrays are expected to increase the number of genes that can be placed on a single chip, allowing for faster diagnosis.
The oncology segment is expected to be the most prominent market segment based on the application. Hereditary cancer abnormalities are now primarily diagnosed via mutation analysis. Rigorous medical supervision and many preventive measures help healthy carriers of cancer-predisposing mutations. Cancer therapy has evolved to include personalized drug selection based on the existence of actionable mutations. Molecular diagnostics has become a vital part of promoting personalized cancer medication.
The infectious disease segment is expected to be the fastest-growing segment in the molecular diagnostics market, based on the application. The infectious diseases segment has seen substantial expansion due to rising demand for PCR tests in COVID-19 tests and vaccine research. Growing instances of infectious diseases will only exacerbate the demand for molecular diagnostics in infectious disease applications.
Based on end-users, the diagnostic centers segment is the largest market segment. Many diagnostic laboratories have quickly adopted PCR tests because of the rising demand for COVID-19 tests. Hospitals have discovered that outsourcing testing to external laboratories is less expensive than doing testing in-house, resulting in the rapid rise of the diagnostic center market.
Based on end-users, the hospitals & clinics segment is predicted to represent the second-largest share of the market. The hospital and clinic market segment is being driven by rising demand for PCR testing for admitting patients and increased investment in hospital and clinical laboratories.
North America (the United States, Canada, and Mexico) will dominate the Molecular Diagnostics market from 2022 to 2027. Favorable government regulations during the pandemic, increased investment in molecular diagnostic test kits and reagents, and greater investment in research and development of molecular diagnostics for various applications in the region have led to the domination of the North American region in the molecular diagnostics market.
The United States is expected to have the lion's share in the North American molecular diagnostics market. This is attributed to the greater adoption of diagnostic tests for oncology applications and heavy investment in PCR-based molecular diagnsotics.
However, the Asia-Pacific region is expected to be the fastest-growing region in the molecular diagnostics market during the forecast period. The rapid growth of the region's pharmaceutical manufacturing industry, increased spending on the healthcare system, and growing acceptance of molecular diagnostic tests are all likely to drive the molecular diagnostics market in the APAC region.
China is expected to hold the largest share of the Asia Pacific molecular diagnostics market. The country's vast population, proactive use of PCR tests to address the need for PCR tests to combat COVID-19 spread, and unmet demand have all contributed to the country's growing molecular diagnostics industry.
Siemens Healthcare GmbH, F. Hoffmann-La Roche Ltd, bioMérieux SA, Laboratory Corporation of America, Quest Diagnostics, Hologic, Inc., Abbott, Becton, Dickinson and Company (BD), Thermo Fisher Scientific Inc., DiaSorin Company, Beckman Coulter Inc., BioFire Defense, LLC, Co-Diagnostics, Inc., Sysmex Corporation, Johnson & Johnson Services, Inc., Hologic Corporation, Illumina Ltd., PCR Biosystems, PCR-Dx, Curiosity Diagnostic, Genome Diagnostics Pvt. Ltd., Molbio Diagnostics Pvt. Ltd., 1drop Inc., Exact Sciences Laboratories, LumiraDx UK Ltd., and Sherlock BioSciences, Inc., among others, are the some of the key players in the molecular diagnostics market.
Please note: This is not an exhaustive list of companies profiled in the report.
Chapter 1 Research Methodology
1.1 Research Assumptions
1.2 Research Methodology
1.2.1 Estimates and Forecast Timeline
1.3 Information Procurement
1.3.1 Purchased Database
1.3.2 GME’s Internal Database
1.3.3 Primary Research
1.3.4 Secondary Sources & Third-Party Perspectives
1.3.4.1 Company Information Sources: Annual Reports, Investor Presentation, Press Release, SEC Filing, Company Blogs & Website
1.3.4.2 Secondary Data Sources: WHO, US FDA, National Cancer Institute, World Bank, IMF
1.4 Information or Data Analysis
1.4.1 Data Analysis Models
1.5 Market Technology & Data Visualization
1.6 Data Validation & Publishing
1.7 Market Model
1.7.1 Model Details
1.7.1.1 Top-Down Approach
1.7.1.2 Bottom-Up Approach
1.8 Market Segmentation & Scope
1.9 Market Definition
Chapter 2 Executive Summary
2.1. Global Market Outlook
2.2 Product Outlook
2.3 Application Outlook
2.4 Technology Outlook
2.5 End-User Outlook
2.6 Regional Outlook
Chapter 3 Global Molecular Diagnostics Market Trend Analysis
3.1. Market Introduction
3.2 Penetration & Growth Prospect Mapping
3.3 Impact of COVID-19 on the Molecular Diagnostics Market
3.4 Metric Data on Food & Beverage Industry
3.5 Market Dynamic
3.5.1 Market Driver Analysis
3.5.2 Market Restraint Analysis
3.5.3 Industry Challenges
3.5.4 Industry Opportunities
3.6 Porter’s Five Analysis
3.6.1 Supplier Power
3.6.2 Buyer Power
3.6.3 Substitution Threat
3.6.4 Threat from New Entrant
3.7 Market Entry Strategies
Chapter 4 Molecular Diagnostics Market: Product Trend Analysis
4.1 Product: Historic Data vs. Forecast Data Analysis, 2021 vs. 2027
4.2 Instruments
4.2.1 Market Estimates & Forecast Analysis of Instruments Segment, By Region, 2019-2027 (USD Billion)
4.3 Consumables
4.3.1 Market Estimates & Forecast Analysis of Consumables Markets Segment, By Region, 2019-2027 (USD Billion)
Chapter 5 Molecular Diagnostics Market: Application Trend Analysis
5.1 Application: Historic Data vs. Forecast Data Analysis, 2021 vs. 2027
5.2 Infectious Disease
5.2.1 Market Estimates & Forecast Analysis of Infectious Disease Segment, By Region, 2019-2027 (USD Billion)
5.3 Oncology
5.3.1 Market Estimates & Forecast Analysis of Oncology Segment, By Region, 2019-2027 (USD Billion)
5.4 Infectious Diseases
5.4.1 Market Estimates & Forecast Analysis of Infectious Diseases Segment, By Region, 2019-2027 (USD Billion)
5.5 Cardiovascular Diseases
5.5.1 Market Estimates & Forecast Analysis of Cardiovascular Diseases Segment, By Region, 2019-2027 (USD Billion)
5.6 Others
5.6.1 Market Estimates & Forecast Analysis of Others Segment, By Region, 2019-2027 (USD Billion)
Chapter 6 Molecular Diagnostics Market: Technology Trend Analysis
6.1 Technology: Historic Data vs. Forecast Data Analysis, 2021 vs. 2027
6.2 PCR
6.2.1 Market Estimates & Forecast Analysis of PCR Segment, By Region, 2019-2027 (USD Billion)
6.3 In Situ Hybridization
6.3.1 Market Estimates & Forecast Analysis of In Situ Hybridization Segment, By Region, 2019-2027 (USD Billion)
6.4 Chips and Microarrays
6.4.1 Market Estimates & Forecast Analysis of PCR Segment, By Region, 2019-2027 (USD Billion)
6.5 Mass Spectrometry (MS)
6.5.1 Market Estimates & Forecast Analysis of Mass Spectrometry (MS) Segment, By Region, 2019-2027 (USD Billion)
6.6 Sequencing
6.6.1 Market Estimates & Forecast Analysis of Sequencing Segment, By Region, 2019-2027 (USD Billion)
6.7 Other Technologies
6.7.1 Market Estimates & Forecast Analysis of Other Technologies Segment, By Region, 2019-2027 (USD Billion)
Chapter 7 Molecular Diagnostics Market: End-User Trend Analysis
7.1 End-User: Historic Data vs. Forecast Data Analysis, 2021 vs. 2027
7.2 Hospitals & Clinics
7.2.1 Market Estimates & Forecast Analysis of Hospitals & Clinics Segment, By Region, 2019-2027 (USD Billion)
7.3 Laboratories
7.3.1 Market Estimates & Forecast Analysis of Laboratories Segment, By Region, 2019-2027 (USD Billion)
7.4 Diagnostic Centers
7.4.1 Market Estimates & Forecast Analysis of Diagnostic Centers Segment, By Region, 2019-2027 (USD Billion)
7.5 Home Testing
7.5.1 Market Estimates & Forecast Analysis of Home Testing Segment, By Region, 2019-2027 (USD Billion)
7.6 Others
7.6.1 Market Estimates & Forecast Analysis of Others Segment, By Region, 2019-2027 (USD Billion)
Chapter 8 Molecular Diagnostics Market, By Region
8.1 Regional Outlook
8.2 North America
8.2.1 Market Estimates & Forecast Analysis, By Country 2019-2027(USD Billion)
8.2.2 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.2.3 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.2.4 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.2.5 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.2.6 U.S.
8.2.6.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.2.6.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.2.6.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.2.6.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.2.7 Canada
8.2.7.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.2.7.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.2.7.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.2.7.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.2.8 Mexico
8.2.8.1 Market Estimates & Forecast Analysis, By Product, 2019-2027 (USD Billion)
8.2.8.2 Market Estimates & Forecast Analysis, By Application, 2019-2027 (USD Billion)
8.2.8.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027 (USD Billion)
8.2.8.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.3 Europe
8.3.1 Market Estimates & Forecast Analysis, By Country 2019-2027(USD Billion)
8.3.2 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.3.3 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.3.4 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.3.5 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.3.6 Germany
8.3.6.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.3.6.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.3.6.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.3.6.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.3.7 UK
8.3.7.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.3.7.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.3.7.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.3.7.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.3.8 France
8.3.8.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.3.8.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.3.8.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.3.8.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.3.9 Russia
8.3.9.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.3.9.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.3.9.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.3.9.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.3.10 Italy
8.3.10.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.3.10.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.3.10.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.3.10.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.3.11 Spain
8.3.11.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.3.11.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.3.11.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.3.11.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.3.12 Rest of Europe
8.3.12.1 Market Estimates & Forecast Analysis, By Product, 2019-2027 (USD Billion)
8.3.12.2 Market Estimates & Forecast Analysis, By Application, 2019-2027 (USD Billion)
8.3.12.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027 (USD Billion)
8.3.12.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.4 Asia Pacific
8.4.1 Market Estimates & Forecast Analysis, By Country 2019-2027(USD Billion)
8.4.2 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.4.3 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.4.4 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.4.5 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.4.6 China
8.4.6.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.4.6.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.4.6.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.4.6.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.4.7 India
8.4.7.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.4.7.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.4.7.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.4.7.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.4.8 Japan
8.4.8.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.4.8.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.4.8.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.4.8.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.4.9 Australia
8.4.9.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.4.9.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.4.9.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.4.9.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.4.10 South Korea
8.4.10.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.4.10.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.4.10.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.4.10.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.4.11 Rest of Asia Pacific
8.4.11.1 Market Estimates & Forecast Analysis, By Product, 2019-2027 (USD Billion)
8.4.11.2 Market Estimates & Forecast Analysis, By Application, 2019-2027 (USD Billion)
8.4.11.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027 (USD Billion)
8.4.11.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.5 Central & South America
8.5.1 Market Estimates & Forecast Analysis, By Country 2019-2027(USD Billion)
8.5.2 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.5.3 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.5.4 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.5.5 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.5.6 Brazil
8.5.6.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.5.6.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.5.6.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.5.6.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.5.7 Rest of Central & South America
8.5.7.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.5.7.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.5.7.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.5.7.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.6 Middle East & Africa
8.6.1 Market Estimates & Forecast Analysis, By Country 2019-2027(USD Billion)
8.6.2 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.6.3 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.6.4 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.6.5 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.6.6 Saudi Arabia
8.6.6.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.6.6.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.6.6.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.6.6.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.6.7 United Arab Emirates
8.6.7.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.6.7.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.6.7.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.6.7.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.6.8 South Africa
8.6.8.1 Market Estimates & Forecast Analysis, By Product, 2019-2027(USD Billion)
8.6.8.2 Market Estimates & Forecast Analysis, By Application, 2019-2027(USD Billion)
8.6.8.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027(USD Billion)
8.6.8.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
8.6.9 Rest of Middle East & Africa
8.6.9.1 Market Estimates & Forecast Analysis, By Product, 2019-2027 (USD Billion)
8.6.9.2 Market Estimates & Forecast Analysis, By Application, 2019-2027 (USD Billion)
8.6.9.3 Market Estimates & Forecast Analysis, By Technology, 2019-2027 (USD Billion)
8.6.9.4 Market Estimates & Forecast Analysis, By End-User, 2019-2027(USD Billion)
Chapter 9 Competitive Analysis
9.1 Key Global Players, Recent Developments & their Impact on the Industry
9.2 Four Quadrant Competitor Positioning Matrix
9.2.1 Key Innovators
9.2.2 Market Leaders
9.2.3 Emerging Players
9.2.4 Market Challengers
9.3 Vendor Landscape Analysis
9.4 End-User Landscape Analysis
9.5 Company Market Share Analysis, 2021
Chapter 10 Company Profile Analysis
10.1 F. Hoffmann-La Roche Ltd
10.1.1 Company Overview
10.1.2 Financial Analysis
10.1.3 Strategic Initiatives
10.1.4 Product Benchmarking
10.2 Danaher Corporation
10.2.1 Company Overview
10.2.2 Financial Analysis
10.2.3 Strategic Initiatives
10.2.4 Product Benchmarking
10.3 Quest Diagnostics Inc.
10.3.1 Company Overview
10.3.2 Financial Analysis
10.3.3 Strategic Initiatives
10.3.4 Product Benchmarking
10.4 Myriad Genetics
10.4.1 Company Overview
10.4.2 Financial Analysis
10.4.3 Strategic Initiatives
10.4.4 Product Benchmarking
10.5 Qiagen NV
10.5.1 Company Overview
10.5.2 Financial Analysis
10.5.3 Strategic Initiatives
10.5.4 Product Benchmarking
10.6 FreeStreamsLive
10.6.1 Company Overview
10.6.2 Financial Analysis
10.6.3 Strategic Initiatives
10.6.4 Product Benchmarking
10.7 Becton, Dickinson and Company
10.7.1 Company Overview
10.7.2 Financial Analysis
10.7.3 Strategic Initiatives
10.7.4 Product Benchmarking
10.8 Abbott Laboratories
10.8.1 Company Overview
10.8.2 Financial Analysis
10.8.3 Strategic Initiatives
10.8.4 Product Benchmarking
10.9 Biomerieux SA
10.9.1 Company Overview
10.9.2 Financial Analysis
10.9.3 Strategic Initiatives
10.9.4 Product Benchmarking
10.10 Bio-Rad Laboratories Inc.
10.10.1 Company Overview
10.10.2 Financial Analysis
10.10.3 Strategic Initiatives
10.10.4 Product Benchmarking
10.11 Sysmex Corporation
10.11.1 Company Overview
10.11.2 Financial Analysis
10.11.3 Strategic Initiatives
10.11.4 Product Benchmarking
10.12 Other Companies
10.12.1 Company Overview
10.12.2 Financial Analysis
10.12.3 Strategic Initiatives
10.12.4 Product Benchmarking
List of Tables
1 Technological Advancements In Molecular Diagnostics Market
2 Global Molecular Diagnostics Market: Key Market Drivers
3 Global Molecular Diagnostics Market: Key Market Challenges
4 Global Molecular Diagnostics Market: Key Market Opportunities
5 Global Molecular Diagnostics Market: Key Market Restraints
6 Global Molecular Diagnostics Market Estimates & Forecast Analysis, 2019-2027 (USD Billion)
7 Global Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
8 Instruments: Global Molecular Diagnostics Market, By Region, 2019-2027 (USD Billion)
9 Consumables Markets: Global Molecular Diagnostics Market, By Region, 2019-2027 (USD Billion)
10 Global Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
11 Infectious Disease: Global Molecular Diagnostics Market, By Region, 2019-2027 (USD Billion)
12 Oncology: Global Molecular Diagnostics Market, By Region, 2019-2027 (USD Billion)
13 Global Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
14 PCR: Global Molecular Diagnostics Market, By Region, 2019-2027 (USD Billion)
15 In Situ Hybridization: Global Molecular Diagnostics Market, By Region, 2019-2027 (USD Billion)
16 Global Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
17 Hospitals & Clinics: Global Molecular Diagnostics Market, By Region, 2019-2027 (USD Billion)
18 Laboratories: Global Molecular Diagnostics Market, By Region, 2019-2027 (USD Billion)
19 Regional Analysis: Global Molecular Diagnostics Market, By Region, 2019-2027 (USD Billion)
20 North America: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
21 North America: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
22 North America: Molecular Diagnostics Market, By Technology , 2019-2027 (USD Billion)
23 North America: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
24 North America: Molecular Diagnostics Market, By Country, 2019-2027 (USD Billion)
25 U.S: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
26 U.S: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
27 U.S: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
28 U.S: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
29 Canada: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
30 Canada: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
31 Canada: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
32 Canada: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
33 Mexico: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
34 Mexico: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
35 Mexico: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
36 Mexico: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
37 Europe: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
38 Europe: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
39 Europe: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
40 Europe: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
41 Germany: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
42 Germany: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
43 Germany: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
44 Germany: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
45 UK: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
46 UK: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
47 UK: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
48 UK: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
49 France: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
50 France: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
51 France: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
52 France: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
53 Italy: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
54 Italy: Molecular Diagnostics Market, By T Application Ype, 2019-2027 (USD Billion)
55 Italy: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
56 ltaly: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
57 Spain: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
58 Spain: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
59 Spain: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
60 Spain: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
61 Rest Of Europe: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
62 Rest Of Europe: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
63 Rest Of Europe: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
64 Rest of Europe: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
65 Asia Pacific: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
66 Asia Pacific: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
67 Asia Pacific: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
68 Asia Pacific: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
69 Asia Pacific: Molecular Diagnostics Market, By Country, 2019-2027 (USD Billion)
70 China: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
71 China: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
72 China: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
73 China: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
74 India: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
75 India: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
76 India: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
77 India: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
78 Japan: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
79 Japan: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
80 Japan: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
81 Japan: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
82 South Korea: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
83 South Korea: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
84 South Korea: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
85 South Korea: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
86 Middle East & Africa: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
87 Middle East & Africa: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
88 Middle East & Africa: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
89 Middle East & Africa: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
90 Middle East & Africa: Molecular Diagnostics Market, By Country, 2019-2027 (USD Billion)
91 Saudi Arabia: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
92 Saudi Arabia: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
93 Saudi Arabia: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
94 Saudi Arabia: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
95 UAE: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
96 UAE: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
97 UAE: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
98 UAE: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
99 Central & South America: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
100 Central & South America: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
101 Central & South America: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
102 Central & South America: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
103 Central & South America: Molecular Diagnostics Market, By Country, 2019-2027 (USD Billion)
104 Brazil: Molecular Diagnostics Market, By Product, 2019-2027 (USD Billion)
105 Brazil: Molecular Diagnostics Market, By Application, 2019-2027 (USD Billion)
106 Brazil: Molecular Diagnostics Market, By Technology, 2019-2027 (USD Billion)
107 Brazil: Molecular Diagnostics Market, By End-User, 2019-2027 (USD Billion)
108 F. Hoffmann-La Roche Ltd: Products Offered
109 Danaher Corporation: Products Offered
110 Quest Diagnostics Inc.: Products Offered
111 Myriad Genetics: Products Offered
112 Qiagen NV: Products Offered
113 FreeStreamsLive: Products Offered
114 Becton, Dickinson and Company: Products Offered
115 Abbott Laboratories: Products Offered
116 Biomerieux SA: Products Offered
117 Bio-Rad Laboratories Inc.: Products Offered
118 Sysmex Corporation: Products Offered
119 Other Companies: Products Offered
List of Figures
1. Global Molecular Diagnostics Market Segmentation & Research Scope
2. Primary Research Partners and Local Informers
3. Primary Research Process
4. Primary Research Approaches
5. Primary Research Responses
6. Global Molecular Diagnostics Market: Penetration & Growth Prospect Mapping
7. Global Molecular Diagnostics Market: Value Chain Analysis
8. Global Molecular Diagnostics Market Drivers
9. Global Molecular Diagnostics Market Restraints
10. Global Molecular Diagnostics Market Opportunities
11. Global Molecular Diagnostics Market Challenges
12. Key Molecular Diagnostics Market Manufacturer Analysis
13. Global Molecular Diagnostics Market: Porter’s Five Forces Analysis
14. PESTLE Analysis & Impact Analysis
15. F. Hoffmann-La Roche Ltd: Company Snapshot
16. F. Hoffmann-La Roche Ltd: Swot Analysis
17. Danaher Corporation: Company Snapshot
18. Danaher Corporation: Swot Analysis
19. Quest Diagnostics Inc.: Company Snapshot
20. Quest Diagnostics Inc.: Swot Analysis
21. Myriad Genetics: Company Snapshot
22. Myriad Genetics: Swot Analysis
23. Qiagen NV: Company Snapshot
24. Qiagen NV: Swot Analysis
25. FreeStreamsLive: Company Snapshot
26. FreeStreamsLive: Swot Analysis
27. Becton, Dickinson and Company: Company Snapshot
28. Becton, Dickinson and Company: Swot Analysis
29. Abbott Laboratories: Company Snapshot
30. Abbott Laboratories: Swot Analysis
31. Biomerieux SA: Company Snapshot
32. Biomerieux SA: Swot Analysis
33. Enchandia AB Software: Company Snapshot
34. Enchandia AB Software: Swot Analysis
35. Bio-Rad Laboratories Inc.: Company Snapshot
36. Bio-Rad Laboratories Inc.: Swot Analysis
37. Sysmex Corporation: Company Snapshot
38. Sysmex Corporation: Swot Analysis
39. Other Companies: Company Snapshot
40. Other Companies: Swot Analysis
The Global Molecular Diagnostics Market has been studied from the year 2019 till 2027. However, the CAGR provided in the report is from the year 2022 to 2027. The research methodology involved three stages: Desk research, Primary research, and Analysis & Output from the entire research process.

The desk research involved a robust background study which meant referring to paid and unpaid databases to understand the market dynamics; mapping contracts from press releases; identifying the key players in the market, studying their product portfolio, competition level, annual reports/SEC filings & investor presentations; and learning the demand and supply-side analysis for the Molecular Diagnostics Market.
The primary research activity included telephonic conversations with more than 50 tier 1 industry consultants, distributors, and end-use product manufacturers.
Finally, based on the above thorough research process, an in-depth analysis was carried out considering the following aspects: market attractiveness, current & future market trends, market share analysis, SWOT analysis of the company and customer analytics.

Frequently Asked Questions
Tailor made solutions just for you
80% of our clients seek made-to-order reports. How do you want us to tailor yours?
OUR CLIENTS