
Chronic Total Occlusion Devices Market Size & Analysis - Forecasts To 2025 By Equipment (Microcatheters, Guidewires, Crossing Devices, Re-Entry Devices, Others), By End-Use (Hospitals & Diagnostic Centers, Ambulatory Care Centers (ACC), Others), By Region (North America, Europe, Asia Pacific, Central & South America, Middle East & Africa), End-Use Landscape, Vendor Landscape, Market Share & Competitive Landscape - Global Market Estimates
Chronic Total Occlusion Devices: Market Insights
The chronic total occlusion devices market is projected to grow at a high CAGR over the coming years on account of increasing demand for treating cardiovascular disease. Chronic total occlusion (CTO) is prevalent in about one-third of patients who undergo angiography. It is expected that the number of bypass surgeries are expected to reduce if surgeons focus on improving their ability to treat CTO.
The growing demand for interventional cardiology devices is also projected to positively influence market growth. Coronary artery disease (CAD) is one of the most common. The increasing number of patients with coronary artery disease (CAD) and peripheral artery disease (PAD) is projected to augment the use of CTO treatment across the world.
Chronic Total Occlusion Devices Market: Equipment and End-Use Segmentation
On the basis of equipment, the chronic total occlusion devices market is segmented into Microcatheters, Guidewires, Crossing Devices, and Re-Entry Devices among others. The chronic total occlusion devices market has been segmented into Hospitals & Diagnostic Centers, Ambulatory Care Centers (ACC), Others; on the basis on end-use.
Chronic Total Occlusion Devices Market: Regional Insights
The demand for chronic total occlusion devices is expected to grow at a high CAGR in North America, Europe and Asia Pacific. Training initiatives taken up by manufactures & associations is expected to benefit physicians in understanding the techniques and technology used in CTO procedure.
Technological developments and improvement in techniques has increased the success rate of CTO percutaneous coronary intervention. Approximately 20% of patients diagnosed with coronary disease on angiography have chronic total occlusion. Increasing investments in healthcare infrastructure in Asia Pacific is expected to positively influence market growth over the coming years. In May 2017, a European randomized trial was conducted to compare PCI with optimized medical therapy. This study revealed high success rate and low procedural risk with PCI, which is expected to increase the preference for CTO devices over the coming years.
Chronic Total Occlusion Devices Market: Vendor Landscape
The report contains a chapter dedicated to vendors operating in the chronic total occlusion devices market, covering raw material manufactures, equipment developers, manufacturers, and distributors. The report provides these insights on a regional level. This section of the report entails contact details, experience, products manufactured/supplied, and geographical presence of companies.
Chronic Total Occlusion Devices Market: End-Use Landscape
The report provides a detailed list of end-users operating across the world. The end-user landscape includes consumer contact details, geographical presence, revenue, product portfolio, organic and inorganic growth strategies among others. Some of the companies who are likely to use chronic total occlusion devices include Wellspan York Hospital, Beth Israel Deaconess Medical Center, and NewYork–Presbyterian Hospital among others.
Chronic Total Occlusion Devices Market: Market Share & Competitor Analysis
Some of the major companies engaged in the chronic total occlusion devices market are Medtronic, Boston Scientific Corporation, Terumo Europe, Cordis Corporation, Spectranetics, IntraLuminal Therapeutics, Bard Peripheral Vascular, Baylis Medical Company, Inc., and Soundbite Medical Solutions among others. The market is expected to witness launch of new products and increased investments for research & training.
Please note: This is not an exhaustive list of companies profiled in the report.
In June 2015, Cordis Corporation launched ELITECROSS Support Catheter in the U.S. and OUTBACK Elite Re-Entry Catheter in the U.S., Europe, and Japan. The products are designed to support physicians in treating complex lesions using interventional techniques. The development of new and improved products is expected to increase the success rate of CTO treatment over the coming years.
Check the Press Release on Chronic Total Occlusion Devices Market Report
We value your investment and offer free customization with every report to fulfil your exact research needs.
The global Chronic Total Occlusion Devices Market has been studied from the year 2016 till 2025. However, the CAGR provided in the report is from the year 2017 to 2025. The research methodology involved three stages: Desk research, Primary research, and Analysis & Output from the entire research process.
The desk research involved a robust background study which meant referring to paid and unpaid databases to understand the market dynamics; mapping contracts from press releases; identifying the key players in the market, studying their product portfolio, competition level, annual reports/SEC filings & investor presentations; and learning the demand and supply side analysis for the Chronic Total Occlusion Devices Market.
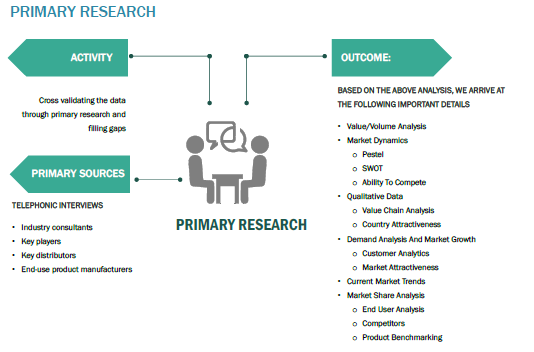
The primary research activity included telephonic conversations with more than 50 tier 1 industry consultants, distributors, and end-use product manufacturers.
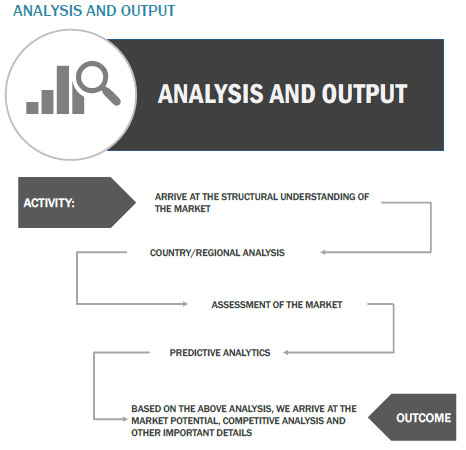
Finally, based on the above thorough research process, an in-depth analysis was carried out considering the following aspects: market attractiveness, current & future market trends, market share analysis, SWOT analysis of the companies, and customer analytics.

Tailor made solutions just for you
80% of our clients seek made-to-order reports. How do you want us to tailor yours?
OUR CLIENTS