
Global Clinical Trial Management System Market Size, Trends & Analysis - Forecasts to 2026 By Deployment Type (Enterprise-Wide CTMS, On-Site CTMS), By Delivery (Web-Based(On-Demand), Licensed Enterprise(On-Premise), Cloud- Based (SaaS)), By Product & Service (Software, Services), By End-User (Large Pharma-Biotech Companies, CROs, Medical Device Manufacturers, Small &Medium Sized-Biotech Companies, Other End Users), By Region (North America, Asia Pacific, Europe, Central & South America, Middle East & Africa); End-User Landscape, Company Market Share Analysis, and Competitor Analysis
The ongoing pandemic has enhanced the need to study the origin of diseases and developing certain vaccines. Clinical trials add value to medical research/knowledge. The system that manages and organizes clinical trials is precisely termed CTMS. This management system is a group of tools that offers the benefits of accessing the latest information, help to effectively collaborate among teams, and provide transparency across multiple documents.
As clinical trials increase worldwide, the need to better access the old data, tracking payments, cost incurred in the clinical trial, and managing the workflow also increases. Software application reduces the burden on the individuals and frees up the resources for other projects.
The software tools for clinical trials are replacing the excel sheets as the clinical research paces towards more complexity and regulatory compliance in the market space. Clinical trials generate high-cost variables from getting patients to volunteer for the trials, recruiting the trained scientists to manage the trials, and using the diagnostic tools/equipment. Much research points towards the high cost incurred in phase 1 trials. The use of software/management system in the clinical study provides cost recovery analysis, history/pattern of the patient volunteer, and the staff capacity needed to maximize the trials.
The COVID-19 pushed the biotechnology and healthcare sector to change drastically. The importance of EHR increased multifold, virtual home visits became a norm, and software applications domain saw high deployment rate. The clinical trials conducted surged in 2020 as companies like Moderna and Pfizer engaged in clinical trials to develop vaccines for the novel virus.
The market’s performance heavily relies on the digital technology invasion. The penetration of wearable devices, AI, and cloud technology have magnified the data collection frequency throughout all phases of trials. Modern software also helps the market to innovate with the trial structures, lead to increased patient satisfaction and implement novel data points in clinical research.
Given the role of companies operating in this space, the clinical trials domain is accentuated by the acquisition, changing market structure, and structured role of IT at a large scale. The market attention is diverted towards the integration of AI into the CTMS. AI can lead the role of a smart assistant in predicting outcomes and automating daily tasks.
Investment in software tools is at an all-time high owing to the manufacturers’ effort to constantly engage in funding rounds with potential investors. The funding helps the market to achieve high product development and better serve the customers. Manufacturers such as eclinical solutions are also actively participating in the investment rounds to expand their product portfolio. Summit Partners and Mainsail Partners are investing heavily in clinical software to strengthen the market capabilities. Other investors enhancing the market potential are Mercia Fund Managers. In 2018, Pharmaseal International secured funding from Mercia Fund Managers to fulfill the rising demand for clinical software.
Medable Inc was also successful in raising the necessary funds to conduct clinical trials to suffice for the rising demand for the same in the COVID-19. The funding and rising investment in the market raise the market’s expansionary capabilities.
The enterprise-wide type would bear a huge market dominance due to the collective efforts of the organization/ universities to effectively deploy it. The University of Minnesota has been successful in the past in the deployment of enterprise-wide type software. The university aimed to support the research in oncology, cardiology, and pediatric medicine. The importance of CTMS for the campus-wide implementation was to assist pharmaceutical and cancer trials, and multi-dimensional studies. IMPACT CTMS is an example of an enterprise-wide type that is used by top companies in the pharmaceutical domain.
The cloud-based domain would have a huge market undertaking due to the varied benefits the unit offers. Cloud-based effectively reduces the organization’s need to maintain IT services. The segment enables the easy accessibility of data at any point of location and time, has the feature to innovate rapidly, and leads to less time spent on upgrading the system.
The software would serve as the largest market driver component. The ability of the software to magnify the market performance has been applauded greatly and accepted across multiple domain-biotechnology, pharma, and healthcare. The exploitation of software by the CROs leads to a centralized system of the database. The updated database helps to better serve the customers through increased transparency and control.
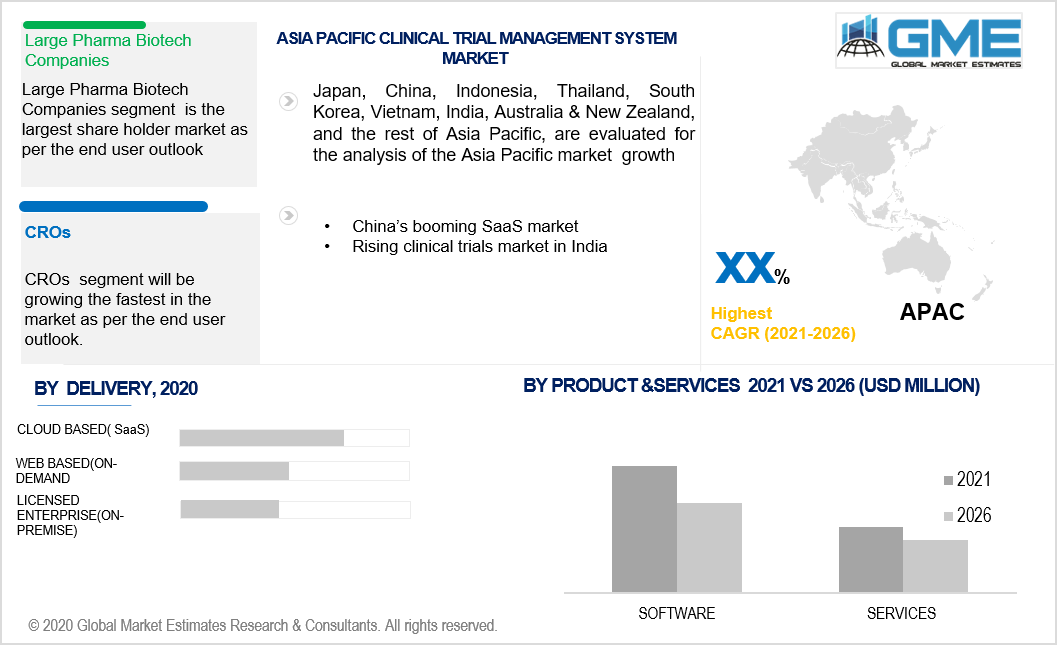
CROs as the end-user would be claiming a large market space. The rising inclination towards the transfer of clinical trial studies to the CROs would have a huge bearing on the use of CTMS by the CROs. The Life Sciences industry is actively participating in outsourcing major clinical work to CROs, increasing the CROs’ role in the market space, and use of CTMS from their end.
Large enterprises would position themselves as a dominant leader owing to the large-scale use of CTMS by the universities, global pharma players and medical instates to integrate an efficient partnership network of teams.
North America’s dominating position would be strengthened by the rising number of clinical trials and the presence of regulatory authority to approve timely clinical trials. The growing expenditure in the pharmaceutical domain would also work to benefit the market in the region. The growing nature of the pharmaceutical partnerships also has a large bearing on the market demand.
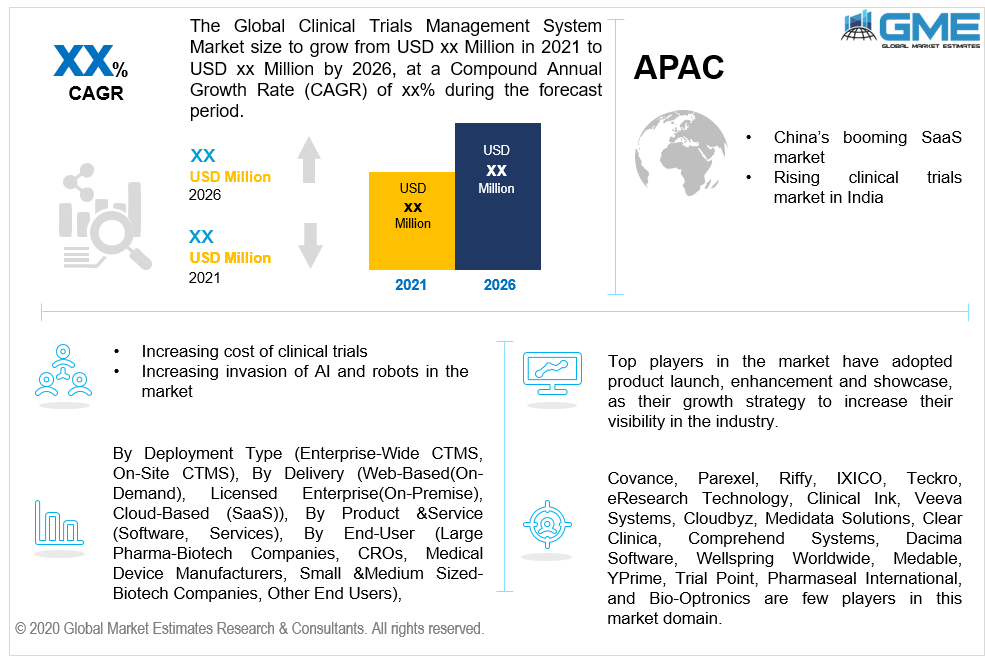
Asia Pacific’s emerging position in this market is sustained by the increasing number of software companies and clinical trials. India’s strong position in the clinical trial domain will have a strong bearing on the region’s rising capabilities to support this market. China’s emerging SaaS industry also enhances the market’s potential in the region
Covance, Parexel, Riffy, IXICO, Teckro, eResearch Technology, Clinical Ink, Veeva Systems, Cloudbyz, Medidata Solutions, Clear Clinica, Comprehend Systems, Dacima Software, Wellspring Worldwide, Medable, YPrime, Trial Point, Pharmaseal International, and Bio-Optronics are few players in this market domain.
Please note: This is not an exhaustive list of companies profiled in the report.
In October 2020, LabCorp planned to term Covance as a decentralized CRO.
In July 2020, Covance entered into a strategic agreement to acquire GlobalCare.
We value your investment and offer free customization with every report to fulfil your exact research needs.
The Global Clinical Trial Management System Market has been studied from the year 2019 till 2026. However, the CAGR provided in the report is from the year 2021 to 2026. The research methodology involved three stages: Desk research, Primary research, and Analysis & Output from the entire research process.

The desk research involved a robust background study which meant referring to paid and unpaid databases to understand the market dynamics; mapping contracts from press releases; identifying the key players in the market, studying their product portfolio, competition level, annual reports/SEC filings & investor presentations; and learning the demand and supply-side analysis for the Clinical Trial Management System Market.
The primary research activity included telephonic conversations with more than 50 tier 1 industry consultants, distributors, and end-use product manufacturers.
Finally, based on the above thorough research process, an in-depth analysis was carried out considering the following aspects: market attractiveness, current & future market trends, market share analysis, SWOT analysis of the company and customer analytics.

Tailor made solutions just for you
80% of our clients seek made-to-order reports. How do you want us to tailor yours?
OUR CLIENTS