
Global Medical Device Testing Market Size, Trends, and Analysis - Forecasts To 2026 By Testing (Electrical Safety, Cybersecurity Testing, Mechanical Testing, Electromagnetic Compatibility, Environmental Testing, Chemical, Physical and Biocompatibility Testing), By Service Type (Testing Services, Certification Services, Inspection Services, Others), By Sourcing Type (In-House, Outsourced), By Device Class (Class I, Class II, Class III), By Technology (Active Implant Medical Device, Active Medical Device, Non-Active Medical Device, In-Vitro Diagnostic Medical Device, Ophthalmic Medical Device, Orthopedic Medical Device, Vascular Medical Device, Other Medical Device Technologies), By Region (North America, Asia Pacific, CSA, Europe, and the Middle East and Africa); End-User Landscape, Company Market Share Analysis & Competitor Analysis
Testing of medical devices is the method of showing that the system works effectively and safely in operation. It is a crucial step in turning a revolutionary concept into a product that is effective and highly marketable. This is a standard test conducted by government-certified organizations to check whether the existing safety and performance criteria can be met by a medical device. The rising need for validation and verification of medical devices is driving the growth of the testing market for medical devices. Another aspect that will further fuel market development is rising regulatory legislation to maintain the high quality of the product. The major factors driving the growth of the market are increasingly focusing on real-time patient tracking combined with the production of innovative low-cost medical devices. Furthermore, the rising advancement of artificial intelligence and IoT in multiple medical devices and the ongoing prevalence of outsourcing medical device testing services are driving the market growth.
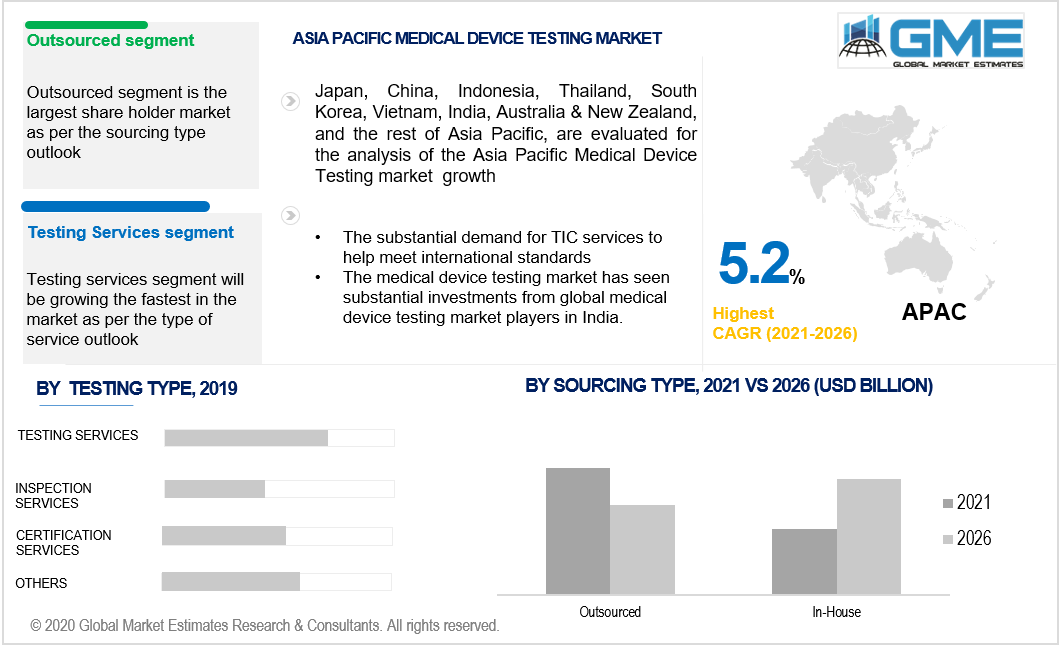
The different services are examination services, certification services, and inspection services, among others, depending on the service viewpoint. Testing services are estimated to retain, in terms of value, the largest market shareholder in 2019. Due to the growing demand for high quality and standard products across the sector, there is a rising demand for TIC services in medical equipment. In laboratories and research centers, testing services are carried out. Testing services ensure that the goods have met the appropriate quality, safety, and performance regulatory requirements in the medical device testing industry.
Outsourcing will expand at a high CAGR throughout the predicted period by sourcing form. Due to the capital-intensive nature of in-house TIC operations, demand for outsourcing TIC services to third-party vendors is growing for technologies like active implant medical devices. Moreover, with large numbers of small and mid-sized players, the overall outsourced medical device testing sector is moderately oriented.
The class III device category is projected to dominate the market at a high CAGR during the forthcoming years. Class III devices are high-risk devices and are thus subject to the highest regulatory control level. To sustain or help human life, Class III medical devices are used. Owing to the presence of possible hazards related to medical devices and affected patients, there is a growing need to identify medical devices.
An in-vitro diagnostic medical device is projected to hold the largest market shareholder in 2019 in terms of value. It contributes enormous value to the process of treatment and medical diagnosis, which increases the well-being of public health at the same time. Such tools help identify infections, diagnose medical problems, and monitor drug treatments. Furthermore, due to its direct effect on the safety and health of workers, active implant medical device technology continues to develop at a higher CAGR during the predicted time.
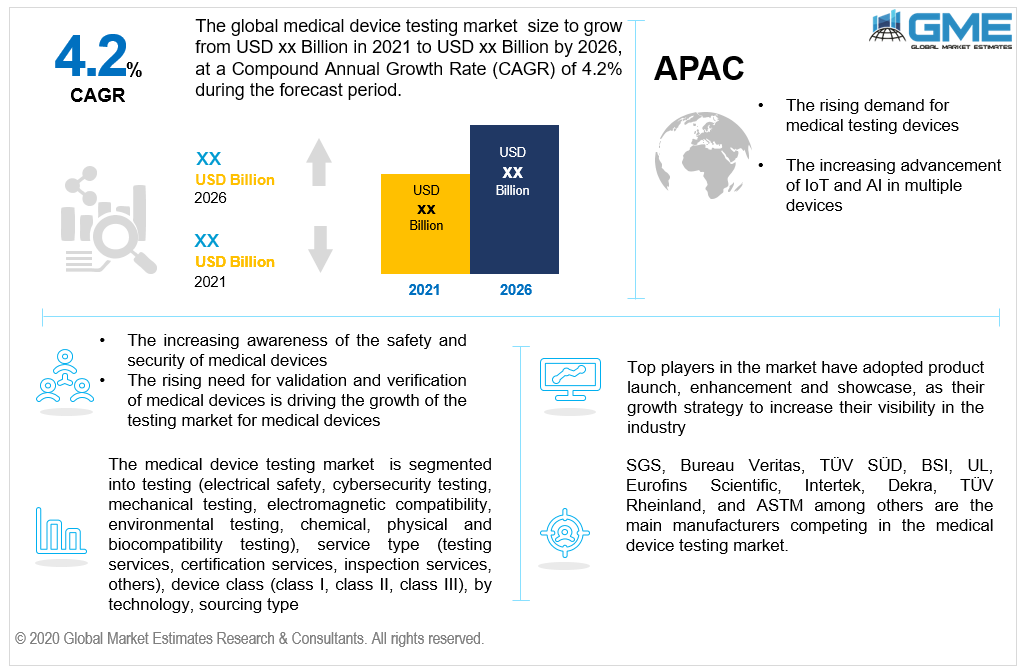
The North American region occupies the leading market segment in terms of revenue over the coming years. The main factors driving this regional market are expected to be increasing complexity in product design and increasing initiatives towards cost-cutting. Moreover, increasing awareness of the safety and security of medical devices is one of the key indicators driving the market growth in the region. Furthermore, as a leading producer of medical device testing, the Asia Pacific region is projected to be the fastest-growing segment due to the availability of skilled labour at lower costs. The increasing consumer awareness of the significance of certification is driving the growth of the region's market.
SGS, Bureau Veritas, TÜV SÜD, BSI, UL, Eurofins Scientific, Intertek, Dekra, TÜV Rheinland, and ASTM among others are the main manufacturers competing in the medical device testing market.
Please note: This is not an exhaustive list of companies profiled in the report.
In June 2020, Intertek announced the extension of its private protective equipment services to include N95 respirator pre-certification testing to the standards set by the National Institute for Occupational Safety and Health (NIOSH)
We value your investment and offer free customization with every report to fulfil your exact research needs.
The Global Medical Device Testing Market has been studied from the year 2019 till 2026. However, the CAGR provided in the report is from the year 2021 to 2026. The research methodology involved three stages: Desk research, Primary research, and Analysis & Output from the entire research process.

The desk research involved a robust background study which meant referring to paid and unpaid databases to understand the market dynamics; mapping contracts from press releases; identifying the key players in the market, studying their product portfolio, competition level, annual reports/SEC filings & investor presentations; and learning the demand and supply side analysis for the Medical Device Testing Market.
The primary research activity included telephonic conversations with more than 50 tier 1 industry consultants, distributors, and end-use product manufacturers.
Finally, based on the above thorough research process, an in-depth analysis was carried out considering the following aspects: market attractiveness, current & future market trends, market share analysis, SWOT analysis of the companies and customer analytics.

Tailor made solutions just for you
80% of our clients seek made-to-order reports. How do you want us to tailor yours?
OUR CLIENTS