
Global Non-invasive Prenatal Testing Market Size, Trends, and Analysis - Forecasts To 2026 By Gestation Period (0-12 Weeks, 13-24 Weeks, and 25-36 Weeks), By Risk Type (High & Average Risk and Low Risk), By Method (Ultrasound Detection, Biochemical Screening Tests, and Cell-Free DNA in Maternal Plasma Tests), By Technology (NGS, Array Technology, PCR, and Other Technologies), By Product (Consumables & Reagents and Instruments), By End-User (Hospitals & Clinics and Diagnostic Laboratories), By Region (North America, Asia Pacific, CSA, Europe, and the Middle East and Africa); End-User Landscape, Company Market Share Analysis & Competitor Analysis
Prenatal tests are carried out during pregnancy to diagnose the baby for birth defects, many of which are genetic disorders. Non-invasive prenatal testing is carried out by analyzing the tiny fragments of DNA present in the pregnant woman’s bloodstream. Generally, DNA is found in the cell’s nucleus but these fragments of DNA are not enclosed within the cell walls. During pregnancy, DNA from the fetus and the mother is present in the bloodstream as the fetus’s DNA is carried out into the mother’s blood through the placenta.
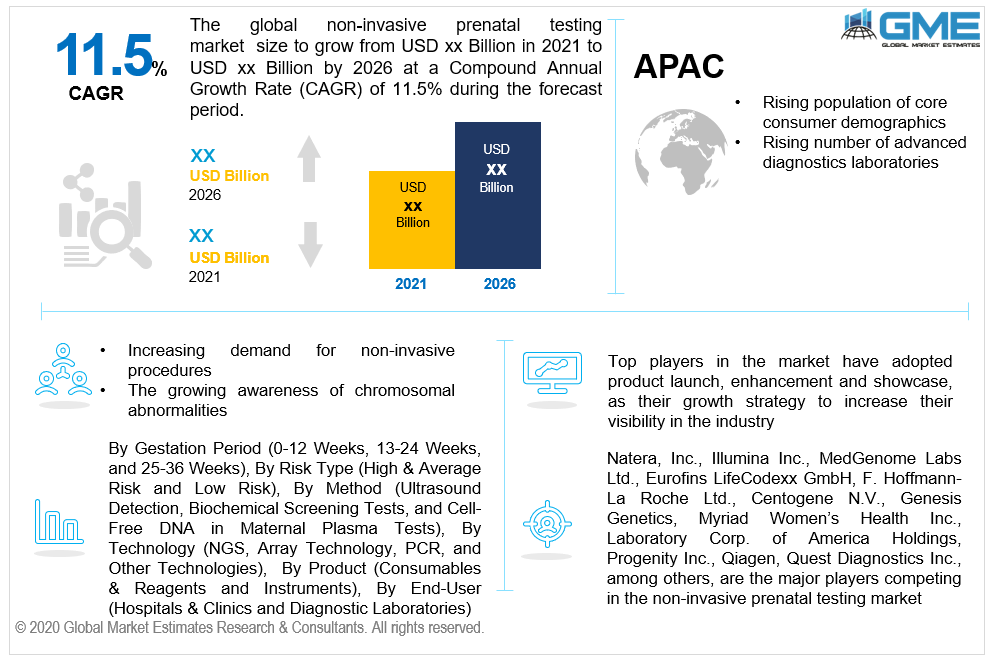
Non-invasive prenatal testing is often regarded as NIPT. NIPT can be carried out to diagnose chromosomal abnormalities like Down syndrome, trisomy 18 and trisomy 13, and extra or missing X or Y chromosomes. Non-invasive pregnancy examinations are being used to determine the likelihood of a genetic disorder in the fetus. The non-invasive prenatal testing market is driven by the growing instances of chromosomal abnormalities in babies and the new technology being developed in diagnostic imaging. Growing insurance coverage for average and low-risk pregnancies is also expected to have a benefit the non-invasive prenatal testing market.
Technological advancements and their growing implementation like bioinformatics and improved functionality of existing tests are also increasing the demand for non-invasive prenatal testing. The non-invasive prenatal testing market is restrained by constraints like this testing procedure can only show the level of risk and therefore can not be used as a stand-alone test to diagnose genetic defects and abnormalities in the fetus. Non-invasive prenatal testing results have a good chance of showing false negatives and positives as the samples have both the mother’s and the baby’s DNA mixed in them.
Numerous firms are actively working to create technologically sophisticated screening methods that are technologically efficient and effective in the early identification of genetic defects in infants. Manufacturers are primarily focusing on the advancement of innovative non-invasive prenatal testing, as well as strategic alliances and agreements, in order to improve their positions in the NIPT market.
The NIPT market is divided into three segments based on the gestation period during which the testing is performed: 0-12 weeks, 13-24 weeks, and 25-36 weeks. The 13-24 weeks segment is expected to hold the dominant market share. The majority of NIPT is performed during the second trimester, in conjunction with other tests such as serum screening and ultrasound. Moreover, the concentration of fetal DNA increases as the gestation period increases. Fetal genome count in the mother’s blood increases from 16 fetal genomes per milliliter to almost 80 genomes per milliliter by the third trimester, thereby making the 13-24 weeks period ideal for non-invasive prenatal testing.
Based on the risk factor of the pregnancy, the NIPT market can be divided into high & average risk and low-risk segments. The high & average risk segment will be the market leader. The increasing number of high-risk pregnancies in women above the age of 35 has resulted in the dominance of this segment. The growing awareness of chromosomal abnormalities and the benefits of an early diagnosis has increased the demand for non-invasive prenatal testing in high and average-risk pregnancies. The low-risk segment is expected to register the fastest growth rate. Growing government support for low-risk pregnancies to conduct non-invasive prenatal testing is expected to increase the demand. Insurance companies are also improving their coverage to include non-invasive prenatal testing for low-risk pregnancies and not just high and average-risk pregnancies. The growing financial support and reimbursement policies are expected to result in a growth of the low-risk segment of the non-invasive prenatal testing market during the forecast period.
The NIPT market can be categorized based on the method used in non-invasive prenatal testing, namely ultrasound detection, biochemical screening tests, and cell-free DNA in maternal plasma tests. The cell-free DNA-based NIPT segment is estimated to be the market leader. The cell-free DNA in the mother’s blood is the most commonly used non-invasive prenatal test given its ease of sample collection and good reliability. These tests are widely being used to forecast the probability of genetic disorders. Recent technological advancements are expected to further increase the efficiency of these tests, making them safer, and deliver better imaging of the fetus. These improvements are predicted to further increase the demand for cell-free DNA-based NIPT.
Depending on the technology used, the NIPT market can be segmented into NGS, array technology, PCR, and other technologies. The NGS segment is expected to be the leader in the market. NGS technology includes whole-exome sequencing, targeted genome sequencing, and whole-genome sequencing of the parental cell-free DNA testing. The benefits of whole-genome sequencing are one of the major drivers of the NGS segment. NGS offers greater efficiency, penetration, and availability, which has increased the demand for NGS.
The NIPT market can be divided into consumables & reagents and instrument segments based on the different NIPT products. The consumables and reagents segment will be the market leader. The growing adoption of NGS technology-based non-invasive prenatal tests has been the major driver of this segment. NGS-based non-invasive prenatal testing requires various reagents and assays in order to do the tests. The large number of vendors that sell these solutions that are used to isolate cell-free DNA from the blood have also contributed to the dominance of the consumables and reagents segment.
The NIPT market can be divided into hospitals & clinics and diagnostic labs based on the different end-users. The market was dominated by diagnostic laboratories. Outsourcing of non-invasive prenatal testing to external laboratories and the growing number of diagnostic laboratories have resulted in the dominance of the diagnostic laboratories segment.
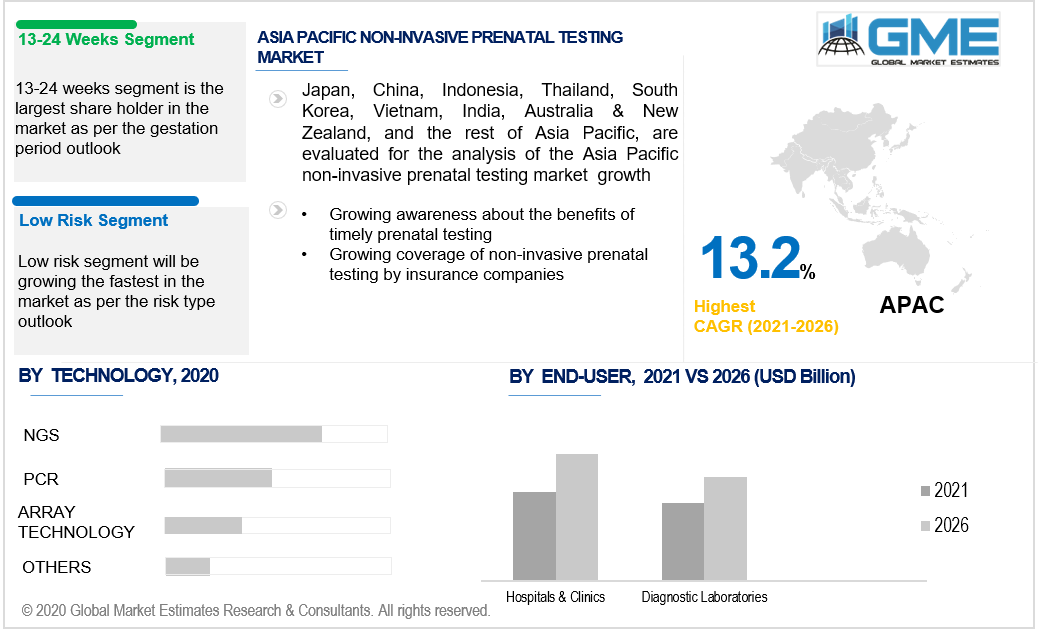
As per the geographical analysis, the market can be classified into North America (the United States, Canada, and Mexico), Asia Pacific (India, China, Japan, Malaysia, Singapore, and Rest of Asia Pacific), Europe (Germany, United Kingdom, Italy, France, Spain, Netherlands, and Rest of Europe), Middle East & Africa (Saudi Arabia, United Arab Emirates, and Rest of the Middle East & Africa) and Central South America (Brazil, Argentina, and Rest of Central and South America). The North American region held the dominant share of the NIPT market. Growing coverage of non-invasive prenatal testing by insurance companies has led to the dominance of this region. The increased healthcare spending and the growing instances of chromosomal abnormalities have been the major drivers of the non-invasive prenatal testing market in the North American region. The Asia Pacific market is predicted to expand at the highest CAGR. The region's demand for non-invasive prenatal testing is projected to rise due to a spike in the number of diagnostic labs and rising population rates.
Natera, Inc., Illumina Inc., MedGenome Labs Ltd., Eurofins LifeCodexx GmbH, F. Hoffmann-La Roche Ltd., Centogene N.V., Genesis Genetics, Myriad Women’s Health Inc., Laboratory Corp. of America Holdings, Progenity Inc., Qiagen, Quest Diagnostics Inc., among others, are the major vendors competing in the non-invasive prenatal testing market.
Please note: This is not an exhaustive list of companies profiled in the report.
We value your investment and offer free customization with every report to fulfil your exact research needs.
The Global Non-invasive Prenatal Testing Market has been studied from the year 2019 till 2026. However, the CAGR provided in the report is from the year 2021 to 2026. The research methodology involved three stages: Desk research, Primary research, and Analysis & Output from the entire research process.

The desk research involved a robust background study which meant referring to paid and unpaid databases to understand the market dynamics; mapping contracts from press releases; identifying the key players in the market, studying their product portfolio, competition level, annual reports/SEC filings & investor presentations; and learning the demand and supply-side analysis for the Non-invasive Prenatal Testing Market.
The primary research activity included telephonic conversations with more than 50 tier 1 industry consultants, distributors, and end-use product manufacturers.
Finally, based on the above thorough research process, an in-depth analysis was carried out considering the following aspects: market attractiveness, current & future market trends, market share analysis, SWOT analysis of the company and customer analytics.

Tailor made solutions just for you
80% of our clients seek made-to-order reports. How do you want us to tailor yours?
OUR CLIENTS