
Global Point Of Care Diagnostic Market Analysis - Forecasts to 2025 By Products (Glucose Testing, Hb1Ac Testing, Coagulation, Fertility, Cardiac Markers, Infectious Diseases, Primary Acre Systems, Hematology, Decentralized Clinical Chemistry, Faeces, Blood Gas Electrolytes, Ambulatory Chemistry, Urinalysis, Drug Abuse Testing), By Prescription Mode (Prescription-Based Testing Kits And Over-The-Counter Testing Kits), By End-Use (Clinics, ,Hospitals, Home, Assisted Living Healthcare Facilities, Laboratory, Emergency Rooms) By Region( North America, Europe, Asia Pacific, Middle East & Africa, Central & South America ), End-use Landscape, Vendor Landscape, & Competitor Analysis | Business Intelligence Report - Global Market Estimates
Market Analysis
The worldwide point of care (PoC) diagnostics market was estimated at USD 23 billion in 2016. The rising prevalence of different chronic and infectious ailments such as heart diseases, hepatitis etc. plays a vital role in the development of the point of care diagnostics products.
The rise in the number of chronic diseases, increase in aging population, innovative advancements, increasing awareness, expanding R&D spending and rapid financial investments in the developing countries are the significant variables expected to drive the PoC diagnostics product demand.
Many advances in optical testing of diagnostic specimens have come in the fields of stream cytometry, haematology, and anatomic pathology including computerized pathology. Such applications are aimed at relieving or minimizing biopsy procedures which are provided at a high cost.
The hike in prevalence of diabetes over the globe is anticipated to boost the demand for glucose testing devices over the years to come. The irresistible illnesses testing packs is expected to develop at the most noteworthy CAGR amid the forecast period on account of the rise in various HIV-infected individuals.
New product launches are one of the primary development strategy embraced by prominent players to expand their portfolio in the global point-of-care diagnostics market. Additionally, strategies such as item endorsements; associations, agreements, and collaborations; and acquisitions were likewise adopted by companies to enhance their product quality and increase their visibility in the market.
Product Insights
The glucose testing segment held the largest share in 2016. Easy availability of PoC glucose checking devices coupled with growing occurrences &pervasiveness of diabetes over the globe are major factors driving the demand of this industry.
Glucose checking gadgets are utilized to gauge the measure of glucose or sugar level in the blood stream. These gadgets provide reliable results, which can be utilized amid normal routine check-ups to interpret risks and furthermore help in the planning of new diet chart & medications.
Increasing utilization of home based glucose checking devices is anticipated to augment demand within the industry. Organizations are expected to launch innovative & advance glucose observing devices which are anticipated to expand offerings to end-users.
Prescription-Modes
OTC testing units are expected to witness higher growth in the market. Prescription based testing units segment holds the largest share in the market which is attributable to rising occurrence and prevalence of lifestyle diseases and absence of awareness on over the counter POCT gadgets.
End-use Insights
Points of care diagnostic devices have observed increased prevalence in hospitals & clinics on account of their affordability and disposability. The contribution of these devices to immediately detect infections and deceases at clinics is also projected to drive demand over the forecast period.
Homecare segment is projected to grow at a high CAGR on account of the convenience these products offer. These products are able to provide results instantly, thereby, giving the consumer sufficient time to address the problem and prevent further health deterioration.
Regional Insights
Various factors such as the developing predominance of way of life ailments, expanding number of item endorsements, and rising government activities for creating novel items in this region are driving the development of the North American market.
The market is fundamentally driven by innovative advancement, cost-effective and high-quality medical solutions. However, the high cost of some procedures and existence of stringent and tedious regulatory framework are expected to hamper market development within the forecast period. Expanding demands from developing economies and health care decentralization may open up new development opportunities in the future.
As indicated by the International Diabetes Federation (IDF), developing countries such as India and China are expected to register the most elevated number of diabetes mellitus patients as compared to developed countries, such as the U.S. and Germany. The count of patients with diabetes mellitus is expected to reach 87 million in India and 62.6 million in China as contrasted with 36 million in the U.S. by 2030.
The Middle Eastern& African market is anticipated to observe huge growth in the near future. Rising demands from the Middle East and African countries such as Saudi Arabia and the UAE are expected to accelerate the demand for point of care testing in the near future. Latin America is also expected to encourage development due to expanding pervasiveness of chronic diseases.
End-use Landscape
The end-use landscape entails a list of current and prospective consumers prevailing across the regions. This section provides company addresses, contact details, products, and regional presence of companies who are purchasing or are likely to purchase PoC devices over the coming years. Some leading purchasers of the PoC diagnostics market are Bumrungrad International Hospital, Palomar Medical Center, and Anadolu Medical Center .
Competitive Scenario
Factors such as developing activities by market players to extend their distribution network, expanding persistent population base, and rising number of organizations and joint ventures among PoC suppliers are expected to drive development in the Asia-Pacific region.
Few of the major players in the test strip market are Roche Diagnostics, Life Scan, Inc., Abbott Laboratories, and Bayer. These players gain dominance over the market as they own state of art manufacturing facilities and strong distribution network.
The new POC troponin T test was launched by Roche Diagnostics in 2015 which has advanced accuracy at low concentrations that helps to detect heart attack patients in hospitals. In 2015, Qualcomm Incorporated (NASDAQ: QCOM), through its subsidiary Qualcomm Life, Inc., entered into an agreement with LifeScan Inc., one of the Johnson & Johnson Diabetes Care Companies (JJDCC), to streamline remote data captured from Life Scan’s OneTouch Verio Flex blood glucose meters to physicians.
1 RESEARCH METHODOLOGY
1.1 DESK RESEARCH
1.2 PRIMARY RESEARCH
1.3 ANALYSIS AND OUTPUT
1.4 FINAL OUTPUT FROM DESK AND PRIMARY RESEARCH
2 ECOSYSTEM OVERVIEW
3 TECHNOLOGICAL TRENDS IN POINT OF CARE DIAGNOSTIC MARKET
4 DEMAND AND SUPPLY MATRIX
4.1 INTRODUCTION
4.2 DEMAND SIDE ANALYSIS
4.2.1 POINT OF CARE DIAGNOSTIC MARKET: MARKET DRIVERS
4.2.2 POINT OF CARE DIAGNOSTIC MARKET: MARKET RESTRAINTS
4.2.3 POINT OF CARE DIAGNOSTIC MARKET: MARKET OPPORTUNITIES
4.2.4 POINT OF CARE DIAGNOSTIC MARKET: MARKET CHALLENGES
4.3 SUPPLY SIDE ANALYSIS
4.3.1 FUTURE SCENARIO (CAGR% VS. COUNTRY)
4.3.2 MAPPING KEY POINT OF CARE DIAGNOSTIC MANUFACTURERS
4.3.3 PORTER’S FIVE FORCES ANALYSIS
4.3.3.1 Threat of new entrants
4.3.3.2 Threat of substitutes
4.3.3.3 Bargaining power of buyers
4.3.3.4 Bargaining power of suppliers
4.3.3.5 Intensity of competitive rivalry
4.3.4 POINT OF CARE DIAGNOSTIC MARKET: TREND ANALYSIS
4.4 PESTLE ANALYSIS
4.5 IMPACT ANALYSIS
4.6 PATENT LISTING
4.7 BEHAVIOURAL ANALYSIS
4.7.1 KEY MACRO INDICATORS
4.7.2 KEY MICRO INDICATORS
5 REGULATORY FRAMEWORK
6 POINT OF CARE DIAGNOSTIC MARKET, BY PRODUCT
6.1 INTRODUCTION
6.2 GLUCOSE TESTING
6.3 HB1AC TESTING
6.4 COAGULATION
6.5 FERTILITY
6.6 CARDIAC MARKERS
6.7 INFECTIOUS DISEASES
6.8 PRIMARY ACRE SYSTEMS
6.9 HEMATOLOGY
6.10 DECENTRALIZED CLINICAL CHEMISTRY
6.11 FAECES
6.12 BLOOD GAS ELECTROLYTES
6.13 AMBULATORY CHEMISTRY
6.14 URINALYSIS
6.15 DRUG ABUSE TESTING
7 POINT OF CARE DIAGNOSTIC MARKET, BY PRESCRIPTION MODE
7.1 INTRODUCTION
7.2 PRESCRIPTION-BASED TESTING KITS
7.3 OVER-THE-COUNTER TESTING KITS
8 POINT OF CARE DIAGNOSTIC MARKET, BY END-USE
8.1 INTRODUCTION
8.2 CLINICS & HOSPITALS
8.3 HOME
8.4 ASSISTED LIVING HEALTHCARE FACILITIES
8.5 LABORATORY
8.6 EMERGENCY ROOMS
9 POINT OF CARE DIAGNOSTIC MARKET, BY REGION
9.1 INTRODUCTION
9.2 NORTH AMERICA
9.2.1 BY PRODUCT
9.2.2 BY PRESCRIPTION MODE
9.2.3 BY APPLICATION
9.2.4 BY COUNTRY
9.2.4.1 U.S.
9.2.4.1.1 By product
9.2.4.1.2 By prescription mode
9.2.4.1.3 By application
9.2.4.2 Canada
9.2.4.2.1 By product
9.2.4.2.2 By prescription mode
9.2.4.2.3 By application
9.3 EUROPE
9.3.1 BY PRODUCT
9.3.2 BY PRESCRIPTION MODE
9.3.3 BY APPLICATION
9.3.4 BY COUNTRY
9.3.4.1 Germany
9.3.4.1.1 By product
9.3.4.1.2 By prescription mode
9.3.4.1.3 By application
9.3.4.2 UK
9.3.4.2.1 By product
9.3.4.2.2 By prescription mode
9.3.4.2.3 By application.
9.3.4.3 France
9.3.4.3.1 By product
9.3.4.3.2 By prescription mode
9.3.4.3.3 By application.
9.3.4.4 Italy
9.3.4.4.1 By product
9.3.4.4.2 By prescription mode
9.3.4.4.3 By application
9.3.4.5 Spain
9.3.4.5.1 By product
9.3.4.5.2 By prescription mode
9.3.4.5.3 By application
9.4 ASIA PACIFIC
9.4.1 BY PRODUCT
9.4.2 BY PRESCRIPTION MODE
9.4.3 BY APPLICATION
9.4.4 BY COUNTRY
9.4.4.1 China
9.4.4.1.1 By product
9.4.4.1.2 By prescription mode
9.4.4.1.3 By application.
9.4.4.2 India
9.4.4.2.1 By product
9.4.4.2.2 By prescription mode
9.4.4.2.3 By application.
9.4.4.3 Japan
9.4.4.3.1 By product
9.4.4.3.2 By prescription mode
9.4.4.3.3 By application.
9.4.4.4 Australia
9.4.4.4.1 By product
9.4.4.4.2 By prescription mode
9.4.4.4.3 By application
9.4.4.5 Singapore
9.4.4.5.1 By product
9.4.4.5.2 By prescription mode
9.4.4.5.3 By application.
9.5 CENTRAL & SOUTH AMERICA
9.5.1 BY PRODUCT
9.5.2 BY PRESCRIPTION MODE
9.5.3 BY APPLICATION
9.5.4 BY COUNTRY
9.5.4.1 BRAZIL
9.5.4.1.1 By product
9.5.4.1.2 By prescription mode
9.5.4.1.3 By application.
9.6 MIDDLE EAST & AFRICA
9.6.1 BY PRODUCT
9.6.2 BY PRESCRIPTION MODE
9.6.3 BY APPLICATION
9.6.4 BY COUNTRY
9.6.4.1 SAUDI ARABIA
9.6.4.1.1 By product
9.6.4.1.2 By prescription mode
9.6.4.1.3 By application.
10 GLOBAL POINT OF CARE DIAGNOSTIC MARKET: END-USER LANDSCAPE (CONSUMER PROFILE)
10.1 INTRODUCTION
10.2 NORTH AMERICA
10.3 EUROPE
10.4 ASIA-PACIFIC
10.5 MIDDLE EAST & AFRICA
10.6 CENTRAL & SOUTH AMERICA
11 VENDOR LANDSCAPE (SUPPLIERS/MANUFACTURERS/DISTRIBUTORS)
11.1 NORTH AMERICA
11.2 EUROPE
11.3 ASIA-PACIFIC
11.4 REST OF THE WORLD
12 COMPANY PROFILE
12.1 ROCHE DIAGNOSTICS
12.1.1 BUSINESS DESCRIPTION
12.1.2 FINANCIAL PERFORMANCE
12.1.3 SWOT ANALYSIS
12.1.4 PRODUCTS AND SERVICES OFFERED
12.1.5 STRATEGIC INITIATIVES
12.2 ABBOTT DIAGNOSTICS INC.
12.2.1 BUSINESS DESCRIPTION
12.2.2 FINANCIAL PERFORMANCE
12.2.3 SWOT ANALYSIS
12.2.4 PRODUCTS AND SERVICES OFFERED
12.2.5 STRATEGIC INITIATIVES
12.3 SIEMENS HEALTHCARE
12.3.1 BUSINESS DESCRIPTION
12.3.2 FINANCIAL PERFORMANCE
12.3.3 SWOT ANALYSIS
12.3.4 PRODUCTS AND SERVICES OFFERED
12.3.5 STRATEGIC INITIATIVES
12.4 DANAHER CORPORATION
12.4.1 BUSINESS DESCRIPTION
12.4.2 FINANCIAL PERFORMANCE
12.4.3 SWOT ANALYSIS
12.4.4 PRODUCTS AND SERVICES OFFERED
12.4.5 STRATEGIC INITIATIVES
12.5 BIOMERIEUX
12.5.1 BUSINESS DESCRIPTION
12.5.2 FINANCIAL PERFORMANCE
12.5.3 SWOT ANALYSIS
12.5.4 PRODUCTS AND SERVICES OFFERED
12.5.5 STRATEGIC INITIATIVES
12.6 ABAXIS, INC.
12.6.1 BUSINESS DESCRIPTION
12.6.2 FINANCIAL PERFORMANCE
12.6.3 SWOT ANALYSIS
12.6.4 PRODUCTS AND SERVICES OFFERED
12.6.5 STRATEGIC INITIATIVES
12.7 ALERE, INC.
12.7.1 BUSINESS DESCRIPTION
12.7.2 FINANCIAL PERFORMANCE
12.7.3 SWOT ANALYSIS
12.7.4 PRODUCTS AND SERVICES OFFERED
12.7.5 STRATEGIC INITIATIVES
12.8 QIAGEN N.V.
12.8.1 BUSINESS DESCRIPTION
12.8.2 FINANCIAL PERFORMANCE
12.8.3 SWOT ANALYSIS
12.8.4 PRODUCTS AND SERVICES OFFERED
12.8.5 STRATEGIC INITIATIVES
12.9 SPECTRAL DIAGNOSTICS
12.9.1 BUSINESS DESCRIPTION
12.9.2 FINANCIAL PERFORMANCE
12.9.3 SWOT ANALYSIS
12.9.4 PRODUCTS AND SERVICES OFFERED
12.9.5 STRATEGIC INITIATIVES
12.10 INSTRUMENTATION LABORATORY
12.10.1 BUSINESS DESCRIPTION
12.10.2 FINANCIAL PERFORMANCE
12.10.3 SWOT ANALYSIS
12.10.4 PRODUCTS AND SERVICES OFFERED
12.10.5 STRATEGIC INITIATIVES
12.11 JOHNSON AND JOHNSON
12.11.1 BUSINESS DESCRIPTION
12.11.2 FINANCIAL PERFORMANCE
12.11.3 SWOT ANALYSIS
12.11.4 PRODUCTS AND SERVICES OFFERED
12.11.5 STRATEGIC INITIATIVES
12.12 NOVA BIOMEDICAL
12.12.1 BUSINESS DESCRIPTION
12.12.2 FINANCIAL PERFORMANCE
12.12.3 SWOT ANALYSIS
12.12.4 PRODUCTS AND SERVICES OFFERED
12.12.5 STRATEGIC INITIATIVES
12.13 NIPRO DIAGNOSTICS INC
12.13.1 BUSINESS DESCRIPTION
12.13.2 FINANCIAL PERFORMANCE
12.13.3 SWOT ANALYSIS
12.13.4 PRODUCTS AND SERVICES OFFERED
12.13.5 STRATEGIC INITIATIVES
12.14 HEMOCUE AB
12.14.1 BUSINESS DESCRIPTION
12.14.2 FINANCIAL PERFORMANCE
12.14.3 SWOT ANALYSIS
12.14.4 PRODUCTS AND SERVICES OFFERED
12.14.5 STRATEGIC INITIATIVES
12.15 INTERNATIONAL TECHNIDYNE CORPORATION (ITC)
12.15.1 BUSINESS DESCRIPTION
12.15.2 FINANCIAL PERFORMANCE
12.15.3 SWOT ANALYSIS
12.15.4 PRODUCTS AND SERVICES OFFERED
12.15.5 STRATEGIC INITIATIVES
The global point of care diagnostics market has been studied from the year 2016 till 2025. However, the CAGR provided in the report is from the year 2017 to 2025. The market research methods involved three stages: Desk research, Primary survey research, and Analysis & Output from the entire research process.
The desk research involved a robust background study which meant referring to paid and unpaid databases to understand the industry analysis; industry trends; market analysis; market trends; market dynamics; mapping contracts from press releases; identifying the key players in the market, studying their product portfolio, competition level, annual reports/SEC filings & investor presentations; and learning the demand and supply side analysis for the point of care diagnostics market.
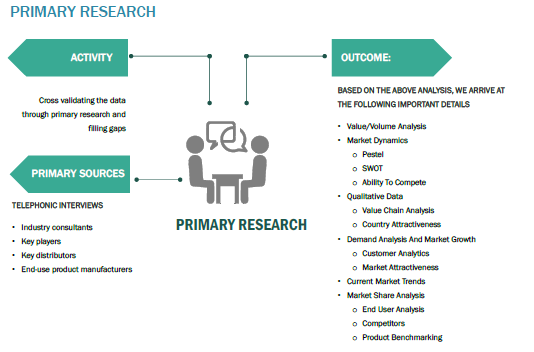
The primary research activity included telephonic conversations with more than 50 tier 1 industry consultants, VPs, CEOs, COOs, financial consultants, business consultants, strategy consultants, project management consultants, business management consultants, distributors, and end-use product manufacturers.
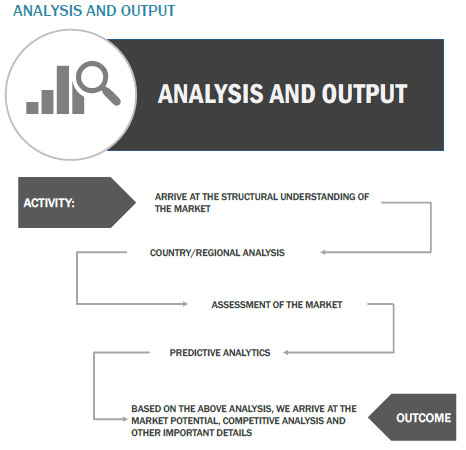
Finally, based on the above thorough research process, an in-depth analysis was carried out considering the following aspects: demand forecasting, market attractiveness, current & future market trends, market share analysis, SWOT analysis of the companies, and customer analytics.

Tailor made solutions just for you
80% of our clients seek made-to-order reports. How do you want us to tailor yours?
OUR CLIENTS