
Global Vaccine Adjuvants Market Size, Trends & Analysis - Forecasts to 2026 By Product Type (Particulate Adjuvants, Adjuvant Emulsions, Pathogen Components, Combination Adjuvants, Others), By Route of Administration (Intramuscular Route, Subcutaneous Route, Intranasal Route, Oral Route, Intradermal Route, Others), By Disease Type (Infectious Diseases, Cancer, Others) By Application (Research Application, Commercial Application), By Application Category (Human Vaccine Adjuvants, Veterinary Vaccine Adjuvants), By Region (North America, Asia Pacific, Europe, Central & South America, Middle East & Africa); End-User Landscape, Company Market Share Analysis, and Competitor Analysis
Adjuvants help the individuals receiving vaccines to develop a strong response to the vaccines. Adjuvants are clinically tested and are under constant clinical trials to increase immunogenicity. Adjuvants are useful in reducing the dose of antigen needed, and also reduce the costs and need for booster shots. Infants receiving the vaccine adjuvants benefit by way of enhanced immune reactions. The use of adjuvants in vaccines has persisted for over 70 years. The efficacy of the adjuvants is predetermined in the clinical trials before the licensing of the vaccines. Continous evaluation, ongoing research are supporting the use of adjuvants in vaccines meant for the mass population.
Citing some of the adjuvants, the use of aluminum salts in vaccines has persisted since the 1930s and it has been used with tetanus and diphtheria vaccines. MF59 is another adjuvant used in the influenza vaccine-Flaud which is licensed to be used in Europe and the USA. The newly manufactured adjuvant CpG 1018 has been used in the Heplisav-B vaccine. This adjuvant has also secured wide-scale investment from top biopharmaceutical players. In 2021, CEPI and Dynavax decided to invest $99 million to increase the manufacturing of CpG 1018. Partnerships and alliances among vaccine developers are also boosting the market demand for Cpg1018 adjuvants. Other adjuvants, AS01?B are also being further clinically tested for the vaccines of HIV and malaria. Manufacturers are also expanding the market for AS01B adjuvant by starting the phase 2a trials for the AS01B adjuvant.
COVID-19 has created a market of its own, as adjuvant CpG1018 is supplied to fulfill the demand for the COVID-19 vaccine. Manufacturers are participating in deals to supply the required adjuvant for the COVID-19 vaccine and also storing it in bulk to helps advance the production of the vaccine for COVID-19. Various studies also point towards the effectiveness of the adjuvants in developing the COVID-19 vaccines.
The market valuation is further enhanced by the research grants provided to accelerate the development of the vaccine adjuvants. Networks like the ENOVA are also shaping the vaccine adjuvant space. ENOVA is a funded program having a wide presence in the EU. EU’s support to the manufacturers is also proving essential for the vaccine adjuvant space. EU in September 2020 extended its support to GSK and Sanofi to meet the region’s demand for adjuvanted vaccine for COVID-19.
The market performance is also analyzed through the view-point of the companies rising in this domain to provide effective vaccines for flu and COVID-19. Amgen, Gilead Sciences, and Altimmune Inc. are few companies that are actively working and partnering up with Universities to expand the vaccine development for COVID-19 and other diseases.
Manufacturers are also scaling up the production of adjuvants through the grants offered. Dynavax in 2020 received a grant of $ 3.4 million to expand the production of CpG1018 to respond to the rising needs for COVID-19 vaccines.
Government subsidies to boost the supply of vaccine adjuvants are also influencing the market performance.
Particulate adjuvant will occupy a significant and dominant market share. The exploitation of particulate adjuvants has been successful to develop an enhanced immune response to fight the poor antigens. Particulate adjuvants have also been developed to support cancer vaccines.
The subcutaneous route remains the ideal choice for the delivery of vaccines by using the syringe and needles. The many benefits associated with this route are that it helps in the longer persistence of drugs.
The intradermal route is being widely used and gaining momentum in the vaccine delivery mechanism. Clinical trials and research are being conducted to develop the efficacy of the intradermal route. The improvements in the intradermal vaccination have been developed to address the challenges of the high price surge scenario for the instance flu pandemic or polio as during these situations the high price of antigen restricts the coverage of the wider population.
Infectious diseases would remain the dominant type as newer vaccine adjuvants are developed to address the rise of infectious diseases. Adjuvants such as QS-21 have been widely investigated and propagated to be used for the treatment of infectious diseases. At present, the adjuvant is investigated in around 15 vaccine samples.
Research application would form the foundation of the application domain. The research on the adjuvants helps in developing improved vaccines, develop and design vaccines for the emergence of new diseases. The research program developed by universities in studying the adjuvants is also being proposed.
The human vaccine adjuvants serve as the established and mature market space. The use of adjuvants in the human vaccine has persisted for more than 90 years. Companies like SEPPIC provide adjuvants to be applied in the human vaccines. AS03 adjuvant has been used in the malaria vaccine and is further developed to be incorporated in human vaccines to treat influenza.
North America’s commanding leadership position is supported by the increased efforts of the countries such as the USA and Canada to regulate and develop adjuvants at a rapid rate. Support from the FDA is also adding to the region’s market demand. The rise of the vaccine developers in the USA to fulfill the demand for COVID-19 vaccines is also boosting the market demand for adjuvants.
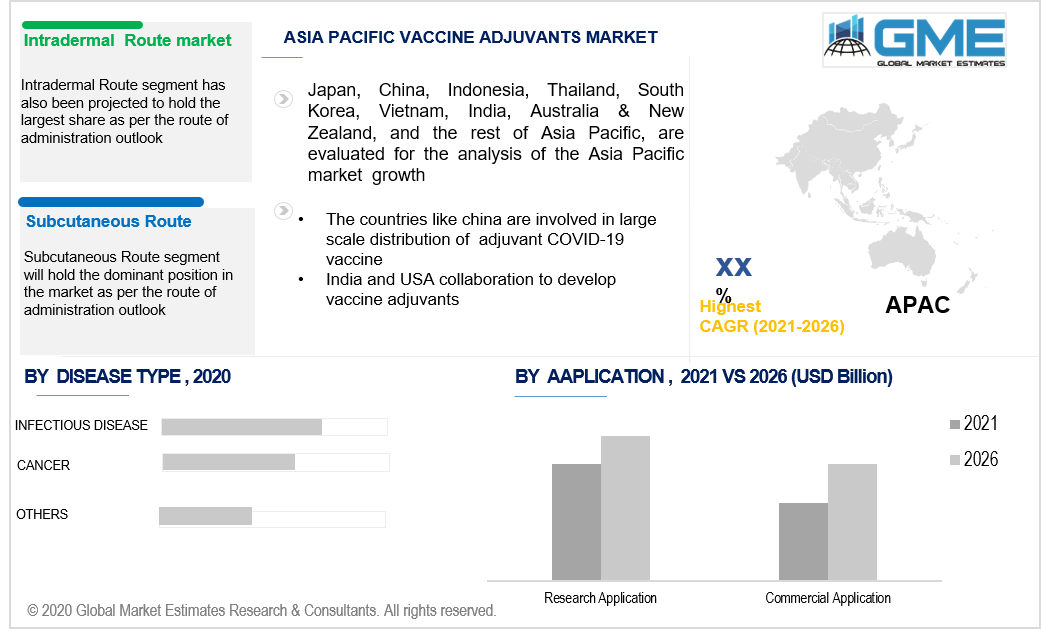
Asia Pacific’s growing potential in the market is derived from the increasing participation of the countries such as India to develop and boost the adjuvant demand. India on the collaborative front is partnering up with the USA to conduct more research on the adjuvants. China is also engaging in the large-scale distribution of vaccine adjuvants to address the spread of COVID-19.
Serum Institute, Glaxo SmithKline, Sinovac Life Sciences, VBI Vaccines, ViroVax, SEPPIC, Invivogen, and Dynavax are key players operating in this market space.
Please note: This is not an exhaustive list of companies profiled in the report.
In October 2020, Bharat Biotech signed a deal with ViraVax to use its adjuvant to enhance the capabilities of Covaxin.
In September 2020, VBI Vaccine partnered with GSK to check the evaluation of cancer vaccine with the AS01B adjuvant.
We value your investment and offer free customization with every report to fulfil your exact research needs.
The Global Vaccine Adjuvants Market has been studied from the year 2019 till 2026. However, the CAGR provided in the report is from the year 2021 to 2026. The research methodology involved three stages: Desk research, Primary research, and Analysis & Output from the entire research process.

The desk research involved a robust background study which meant referring to paid and unpaid databases to understand the market dynamics; mapping contracts from press releases; identifying the key players in the market, studying their product portfolio, competition level, annual reports/SEC filings & investor presentations; and learning the demand and supply-side analysis for the Vaccine Adjuvants Market.
The primary research activity included telephonic conversations with more than 50 tier 1 industry consultants, distributors, and end-use product manufacturers.
Finally, based on the above thorough research process, an in-depth analysis was carried out considering the following aspects: market attractiveness, current & future market trends, market share analysis, SWOT analysis of the company and customer analytics.

Tailor made solutions just for you
80% of our clients seek made-to-order reports. How do you want us to tailor yours?
OUR CLIENTS