
In Vitro Diagnostik Market Analysis - Forecasts to 2025 Technology (Immunodiagnostics, Blood Testing, Molecular Diagnostics, Tissue Diagnostics, Clinical Chemistry, IVD Techniques, Hematology), By Product (Immunochemistry, POCT, Reagents, Software), By Application (Infectious Diseases, Diabetes, Cancer, Cardiac Diseases, Immune System Disorders, Nephrological Diseases, Gastrointestinal Diseases) By End Use (Standalone Laboratory, Hospitals, Academic & Medical Schools, Point of Care Testing), By Region (North America, Europe, Asia Pacific, Central & South America, Middle East & Africa), End-Use Landscape, Vendor Landscape & Competitor Analysis | Business Intelligence Report - Global Market Estimates
Market Analysis
In-vitro diagnostics (IVD) market is expected to enlist a CAGR of more than 5% amid the forecast period. The expanding demand for customized medicine is projected to drive demand in the near future.
Developing healthcare expenses in the rapidly emerging industry, strong demand for technologically propelled testing techniques for diseases diagnosis, and expanding occurrences of chronic diseases are factors expected to augment growth. Operational expansions in clinical research facilities offers scope for advance IVD testing in the developed nations.
IVD plays a vital role in the early detection and management of diseases. The PoC testing industry is developing at a decent pace in the IVD market. The U.S. and Europe dominate the industry in 2016. Growth in these regions is attributed to expanding medicinal expenditures and enhanced clinical laboratory infrastructure.
Technology Insights
In-vitro diagnostics is positively influenced by technological progressions in diagnostic strategies. Analytic arrangements for detecting a large number of diseases are expected to increase the industry’s application scope.
In-vitro diagnostics is utilized for determination of infectious diseases, diabetes, oncology/growth, cardiology ailments, immune system illnesses, nephrology, sedate testing, and HIV/AIDS among others. Growing awareness about medicinal services, demand for cost effective diagnostic solutions, need for speedy and accurate test results are the major factors expected to drive the industry in countries such as India, China, and Japan.
Expanding rates of infectious & chronic diseases, computerized instruments, POC demonstrative arrangements, and increasing government healthcare expenditures are anticipated to augment demand for these services & equipment.
IVD uses therapeutic devices and reagents to analyse samples of blood, tissues, and other body fluids to detect ailments, conditions, and diseases. The tests can be performed in standalone laboratories, hospital labs, and POC testing facilities.
Some of the technological innovations consolidated in in-vitro diagnostics are polymerase chain response (PCR), microarray methods, sequencing and mass spectrometry, which are utilized for test planning. Technological progressions to create high sensitivity POC haematology testing is foreseen to support market development
Product Insights
The immunochemistry segment is a computerized clinical research testing method and records for the large test volume as compared to other methods of testing. The demand for immunochemistry analysers is expected to decline in Europe on account of consolidation of research centres in France and delayed payments received from Italy, Spain, and Portugal.
In China and India, a significant number of rural hospitals need essential diagnostics products and research centre infrastructure. Government funding in the healthcare sectors of these countries is projected to foster demand for immunochemistry analysers over the coming years.
Application Insights
IVD testing is expected to grow at a high CAGR on account of expanding occurrences of chronic & infectious diseases and growing population prone to immunological issues. In addition, rising number of personalized prescriptions and widespread prevalence of uncommon infections are projected to influence the market positively.
The growing prevalence of colorectal tumour, neurological disorders, gynaecological ailments, and others, boosts the acceptance of IVD devices and reagents. On the other hand, stringent government directions and unfavourable repayment arrangements would restrain market development.
The rising demand for technologically advanced diagnostic devices and increasing spending capacity of buyers is likely to give rise to opportunities for IVD development. The demand for customized medication is expanding in the oncology therapeutic area since it encourages better patient management and empowers individualized treatment through diagnostics tests for precise and fast results.
Advancements in the market are expected to drive pharmaceutical organizations to create personalized prescription and offer incentives for cancer treatments. One of the upsides for creating customized treatment is that it empowers better detection of patient analytic data and correspondence results to clinicians.
End-User Insights
The healing centre and academic labs section is relied upon to dominate the market on the grounds, that most indicative tests are performed in-house. The hospitals and analytic research facilities segment held the maximum share in 2016. The demand for healing centres or demonstrative research centres is developing as they provide a proper lab setting, where information obtained from patient samples is used for investigation and development of customized medications.
Regional Insights
North America dominated worldwide in vitro diagnostics industry in 2016. The presence of developed healthcare facilities in the U.S. and Canada, acceptance of customized drug, and growing selection of technologically propelled items is anticipated to augment demand.
The Asia Pacific region is expected to grow at a high CAGR on account of development in research activities and ventures undertaken by companies. Additionally, advancements in new technology and resulting item launches are expected to drive the Asia Pacific market.
End-User Landscape
The end-use landscape entails a list of current and prospective consumers prevailing across the regions. This section provides company addresses, contact details, products, and regional presence of companies who are purchasing or are likely to purchase products from the in-vitro diagnostics market over the coming years. Major players of this market are CTCA, Abbott Laboratories (U.S.), and Valley Cancer Institutes.
Competitor Analysis
The worldwide IVD instruments market is competitive due to the presence of a large number of companies. These vendors collaborate to create advanced products.
Roche Diagnostics and Foundation Medicine teamed entered into a technological collaboration to strengthen the progress of customized treatment for individuals with cancer. Companies have also formed strategic alliances to create & advertise test strips and blood glucose meters.
In 2015 Qualcomm Incorporated (NASDAQ: QCOM), through its subsidiary Qualcomm Life, Inc., entered into an agreement with LifeScan Inc.tolaunch Life Scan’s OneTouch Verio Flex blood glucose meters for physicians which would lead to fast diagnosis and better treatment.
1 RESEARCH METHODOLOGY
1.1 DESK RESEARCH
1.2 PRIMARY RESEARCH
1.3 ANALYSIS AND OUTPUT
1.4 FINAL OUTPUT FROM DESK AND PRIMARY RESEARCH
2 ECOSYSTEM OVERVIEW
3 TECHNOLOGICAL TRENDS IN VITRO DIAGNOSTIK MARKET
4 DEMAND AND SUPPLY MATRIX
4.1 INTRODUCTION
4.2 DEMAND SIDE ANALYSIS
4.2.1 IN VITRO DIAGNOSTIK: MARKET DRIVERS
4.2.2 IN VITRO DIAGNOSTIK: MARKET RESTRAINTS
4.2.3 IN VITRO DIAGNOSTIK: MARKET OPPORTUNITIES
4.2.4 IN VITRO DIAGNOSTIK: MARKET CHALLENGES
4.3 SUPPLY SIDE ANALYSIS
4.3.1 FUTURE SCENARIO (CAGR% VS. COUNTRY)
4.3.2 MAPPING KEY IN VITRO DIAGNOSTIK MANUFACTURERS
4.3.3 PORTER’S FIVE FORCES ANALYSIS
4.3.3.1 Threat of new entrants
4.3.3.2 Threat of substitutes
4.3.3.3 Bargaining power of buyers
4.3.3.4 Bargaining power of suppliers
4.3.3.5 Intensity of competitive rivalry
4.3.4 IN VITRO DIAGNOSTIK MARKET: TREND ANALYSIS
4.4 PESTLE ANALYSIS
4.5 IMPACT ANALYSIS
4.6 PATENT LISTING
4.7 BEHAVIOURAL ANALYSIS
4.7.1 KEY MACRO INDICATORS
4.7.2 KEY MICRO INDICATORS
5 REGULATORY FRAMEWORK
6 IN VITRO DIAGNOSTIK MARKET, BY PRODUCT
6.1 INTRODUCTION
6.2 IMMUNOCHEMISTRY
6.3 POCT
6.4 REAGENTS
6.5 SOFTWARE
7 IN VITRO DIAGNOSTIK MARKET, BY TECHNOLOGY
7.1 INTRODUCTION
7.2 IMMUNODIAGNOSTICS
7.3 BLOOD TESTING
7.4 MOLECULAR DIAGNOSTICS
7.5 TISSUE DIAGNOSTICS
7.6 CLINICAL CHEMISTRY
7.7 IVD TECHNIQUES
7.8 HEMATOLOGY
8 IN VITRO DIAGNOSTIK MARKET, BY APPLICATION
8.1 INTRODUCTION
8.2 INFECTIOUS DISEASES
8.3 DIABETES
8.4 CANCER
8.5 CARDIAC DISEASES
8.6 IMMUNE SYSTEM DISORDERS
8.7 NEPHROLOGICAL DISEASES
8.8 GASTROINTESTINAL DISEASES
9 IN VITRO DIAGNOSTIK MARKET, BY END-USE
9.1 INTRODUCTION
9.2 HOSPITALS
9.3 LABORATORIES
9.4 OTHERS
10 IN VITRO DIAGNOSTIK MARKET, BY REGION
10.1 INTRODUCTION
10.2 NORTH AMERICA
10.2.1 BY PRODUCT
10.2.2 BY TECHNOLOGY
10.2.3 BY APPLICATION
10.2.4 BY END-USE
10.2.5 BY COUNTRY 1
10.2.5.1 U.S.
10.2.5.1.1 By product
10.2.5.1.2 By technology
10.2.5.1.3 By application.
10.2.5.1.4 By end-use
10.2.5.2 CANADA
10.2.5.2.1 By product
10.2.5.2.2 By technology
10.2.5.2.3 By application.
10.2.5.2.4 By end-use
10.2.5.3 MEXICO
10.2.5.3.1 By product
10.2.5.3.2 By technology
10.2.5.3.3 By application.
10.2.5.3.4 By end-use
10.3 EUROPE
10.3.1 BY PRODUCT
10.3.2 BY TECHNOLOGY
10.3.3 BY APPLICATION
10.3.4 BY END-USE
10.3.5 BY COUNTRY
10.3.5.1 GERMANY
10.3.5.1.1 By product
10.3.5.1.2 By technology
10.3.5.1.3 By application.
10.3.5.1.4 By end-use
10.3.5.2 UK
10.3.5.2.1 By product
10.3.5.2.2 By technology
10.3.5.2.3 By application.
10.3.5.2.4 By end-use
10.3.5.3 FRANCE
10.3.5.3.1 By product
10.3.5.3.2 By technology
10.3.5.3.3 By application.
10.3.5.3.4 By end-use
10.3.5.4 ITALY
10.3.5.4.1 By product
10.3.5.4.2 By technology
10.3.5.4.3 By application.
10.3.5.4.4 By end-use
10.3.5.5 SPAIN
10.3.5.5.1 By product
10.3.5.5.2 By technology
10.3.5.5.3 By application.
10.3.5.5.4 By end-use
10.3.5.6 RUSSIA
10.3.5.6.1 By product
10.3.5.6.2 By technology
10.3.5.6.3 By application.
10.3.5.6.4 By end-use
10.4 ASIA PACIFIC
10.4.1 BY PRODUCT
10.4.2 BY TECHNOLOGY
10.4.3 BY APPLICATION
10.4.4 BY END-USE
10.4.5 BY COUNTRY
10.4.5.1 CHINA
10.4.5.1.1 By product
10.4.5.1.2 By technology
10.4.5.1.3 By application.
10.4.5.1.4 By end-use
10.4.5.2 INDIA
10.4.5.2.1 By product
10.4.5.2.2 By technology
10.4.5.2.3 By application.
10.4.5.2.4 By end-use
10.4.5.3 JAPAN
10.4.5.3.1 By product
10.4.5.3.2 By technology
10.4.5.3.3 By application.
10.4.5.3.4 By end-use
10.4.5.4 AUSTRALIA
10.4.5.4.1 By product
10.4.5.4.2 By technology
10.4.5.4.3 By application.
10.4.5.4.4 By end-use
10.4.5.5 SOUTH KOREA
10.4.5.5.1 By product
10.4.5.5.2 By technology
10.4.5.5.3 By application.
10.4.5.5.4 By end-use
10.5 CENTRAL & SOUTH AMERICA
10.5.1 BY PRODUCT
10.5.2 BY TECHNOLOGY
10.5.3 BY APPLICATION
10.5.4 BY END-USE
10.5.5 BY COUNTRY
10.5.5.1 BRAZIL
10.5.5.1.1 By product
10.5.5.1.2 By technology
10.5.5.1.3 By application.
10.5.5.1.4 By end-use
10.6 MIDDLE EAST & AFRICA
10.6.1 BY PRODUCT
10.6.2 BY TECHNOLOGY
10.6.3 BY APPLICATION
10.6.4 BY END-USE
10.6.5 BY COUNTRY
10.6.5.1 SAUDI ARABIA
10.6.5.1.1 By product
10.6.5.1.2 By technology
10.6.5.1.3 By application.
10.6.5.1.4 By end-use
10.6.5.2 U.A.E
10.6.5.2.1 By product
10.6.5.2.2 By technology
10.6.5.2.3 By application.
10.6.5.2.4 By end-use
11 END-USE LANDSCAPE (CONSUMER PROFILE)
11.1 NORTH AMERICA
11.2 EUROPE
11.3 ASIA-PACIFIC
11.4 CENTRAL & SOUTH AMERICA
11.5 MIDDLE EAST & AFRICA
12 VENDOR LANDSCAPE (SUPPLIERS/MANUFACTURERS/DISTRIBUTORS)
12.1 NORTH AMERICA
12.2 EUROPE
12.3 ASIA-PACIFIC
12.4 CENTRAL & SOUTH AMERICA
12.5 MIDDLE EAST & AFRICA
13 COMPANY PROFILE
13.1 ABBOTT LABORATORIES, INC.
13.1.1 BUSINESS DESCRIPTION
13.1.2 FINANCIAL PERFORMANCE
13.1.3 SWOT ANALYSIS
13.1.4 PRODUCTS AND SERVICES OFFERED
13.1.5 STRATEGIC INITIATIVES
13.2 ALERE, INC.
13.2.1 BUSINESS DESCRIPTION
13.2.2 FINANCIAL PERFORMANCE
13.2.3 SWOT ANALYSIS
13.2.4 PRODUCTS AND SERVICES OFFERED
13.2.5 STRATEGIC INITIATIVES
13.3 QIAGEN N.V.
13.3.1 BUSINESS DESCRIPTION
13.3.2 FINANCIAL PERFORMANCE
13.3.3 SWOT ANALYSIS
13.3.4 PRODUCTS AND SERVICES OFFERED
13.3.5 STRATEGIC INITIATIVES
13.4 ROCHE DIAGNOSTICS
13.4.1 BUSINESS DESCRIPTION
13.4.2 FINANCIAL PERFORMANCE
13.4.3 SWOT ANALYSIS
13.4.4 PRODUCTS AND SERVICES OFFERED
13.4.5 STRATEGIC INITIATIVES
13.5 ATLAS GENETICS LTD.
13.5.1 BUSINESS DESCRIPTION
13.5.2 FINANCIAL PERFORMANCE
13.5.3 SWOT ANALYSIS
13.5.4 PRODUCTS AND SERVICES OFFERED
13.5.5 STRATEGIC INITIATIVES
13.6 ENIGMA DIAGNOSTICS, LTD.
13.6.1 BUSINESS DESCRIPTION
13.6.2 FINANCIAL PERFORMANCE
13.6.3 SWOT ANALYSIS
13.6.4 PRODUCTS AND SERVICES OFFERED
13.6.5 STRATEGIC INITIATIVES
13.7 SIEMENS HEALTHCARE
13.7.1 BUSINESS DESCRIPTION
13.7.2 FINANCIAL PERFORMANCE
13.7.3 SWOT ANALYSIS
13.7.4 PRODUCTS AND SERVICES OFFERED
13.7.5 STRATEGIC INITIATIVES
13.8 BECTON, DICKINSON AND COMPANY
13.8.1 BUSINESS DESCRIPTION
13.8.2 FINANCIAL PERFORMANCE
13.8.3 SWOT ANALYSIS
13.8.4 PRODUCTS AND SERVICES OFFERED
13.8.5 STRATEGIC INITIATIVES
13.9 BECKMAN COULTER INC.
13.9.1 BUSINESS DESCRIPTION
13.9.2 FINANCIAL PERFORMANCE
13.9.3 SWOT ANALYSIS
13.9.4 PRODUCTS AND SERVICES OFFERED
13.9.5 STRATEGIC INITIATIVES
13.10 BIOMÉRIEUX SA
13.10.1 BUSINESS DESCRIPTION
13.10.2 FINANCIAL PERFORMANCE
13.10.3 SWOT ANALYSIS
13.10.4 PRODUCTS AND SERVICES OFFERED
13.10.5 STRATEGIC INITIATIVES
13.11 BIO-RAD LABORATORIES, INC.
13.11.1 BUSINESS DESCRIPTION
13.11.2 FINANCIAL PERFORMANCE
13.11.3 SWOT ANALYSIS
13.11.4 PRODUCTS AND SERVICES OFFERED
13.11.5 STRATEGIC INITIATIVES
13.12 THERMO FISHER SCIENTIFIC, INC.
13.12.1 BUSINESS DESCRIPTION
13.12.2 FINANCIAL PERFORMANCE
13.12.3 SWOT ANALYSIS
13.12.4 PRODUCTS AND SERVICES OFFERED
13.12.5 STRATEGIC INITIATIVES
13.13 DANAHER CORPORATION
13.13.1 BUSINESS DESCRIPTION
13.13.2 FINANCIAL PERFORMANCE
13.13.3 SWOT ANALYSIS
13.13.4 PRODUCTS AND SERVICES OFFERED
13.13.5 STRATEGIC INITIATIVES
13.14 JOHNSON & JOHNSON SERVICES, INC.
13.14.1 BUSINESS DESCRIPTION
13.14.2 FINANCIAL PERFORMANCE
13.14.3 SWOT ANALYSIS
13.14.4 PRODUCTS AND SERVICES OFFERED
13.14.5 STRATEGIC INITIATIVES
13.15 SYSMEX CORPORATION
13.15.1 BUSINESS DESCRIPTION
13.15.2 FINANCIAL PERFORMANCE
13.15.3 SWOT ANALYSIS
13.15.4 PRODUCTS AND SERVICES OFFERED
13.15.5 STRATEGIC INITIATIVES
The global In vitro diagnostik market report has been studied from the year 2016 till 2025. However, the CAGR provided in the market study is from the year 2017 to 2025. The market research methods involved three stages: Desk research, Primary survey research, and Analysis & Output from the entire research process.

The desk research involved a robust background study which meant referring to paid and unpaid databases to understand the industry analysis; industry trends; market analysis; market trends; market dynamics; mapping contracts from press releases; identifying the key players in the market, studying their product portfolio, competition level, annual reports/SEC filings & investor presentations; and learning the demand and supply side analysis for the In vitro diagnostik market.
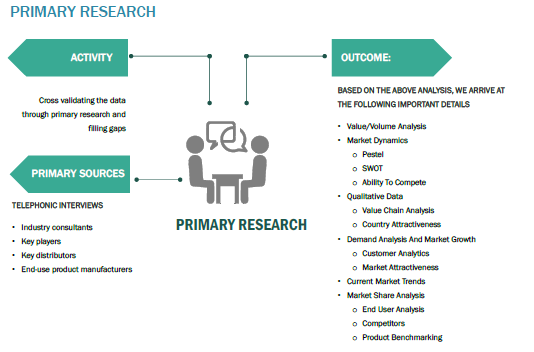
The primary research activity included telephonic conversations with more than 50 tier 1 industry consultants, VPs, CEOs, COOs, financial consultants, business consultants, strategy consultants, project management consultants, business management consultants, distributors, and end-use product manufacturers.
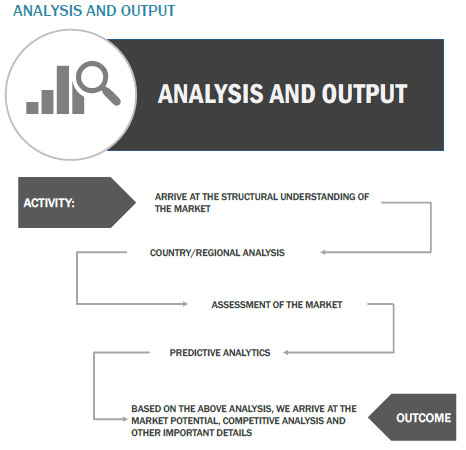
Finally, based on the above thorough research process, an in-depth analysis was carried out considering the following aspects: market attractiveness, demand forecasting, current & future market trends, market share analysis, SWOT analysis of the companies, and customer analytics.

Tailor made solutions just for you
80% of our clients seek made-to-order reports. How do you want us to tailor yours?
OUR CLIENTS